|
|
Post by rockstarrick on Nov 15, 2018 23:52:05 GMT -5
A few weeks ago the deal with UT kept MNKD from running out of money and getting slaughtered in the conference call. Now people are complaining that we should have gotten more money. We shouldn't feel bad about our deal just because another company received a bigger deal. No, but I'm beginning to feel bad that they haven't said anything more about tre-t development. They announce this deal with Arena for a drug 2-3 years or more from approval, and haven't said anything about tre-t despite it being able to go on into phase 3. Meanwhile, Liquidia is progressing steadily towards a late 2019 launch date for its drug. I’m thinking a deal of this magnitude comes with a mandatory PR, which is what was announced today. We will hear news on P3 trial results, NDA, (if it is required for an approved drug that is being delivered by an approved delivery device and Technology), maybe an FDA Avisory Committee Meeting prior to approval, or a dreaded CRL. It would be nice to get at least a few PR’s that aren’t required, but I wouldn’t count on it |
|
|
|
Post by awesomo on Nov 16, 2018 10:15:45 GMT -5
|
|
|
|
Post by harryx1 on Nov 16, 2018 10:52:26 GMT -5
from finsel on stocktwits... 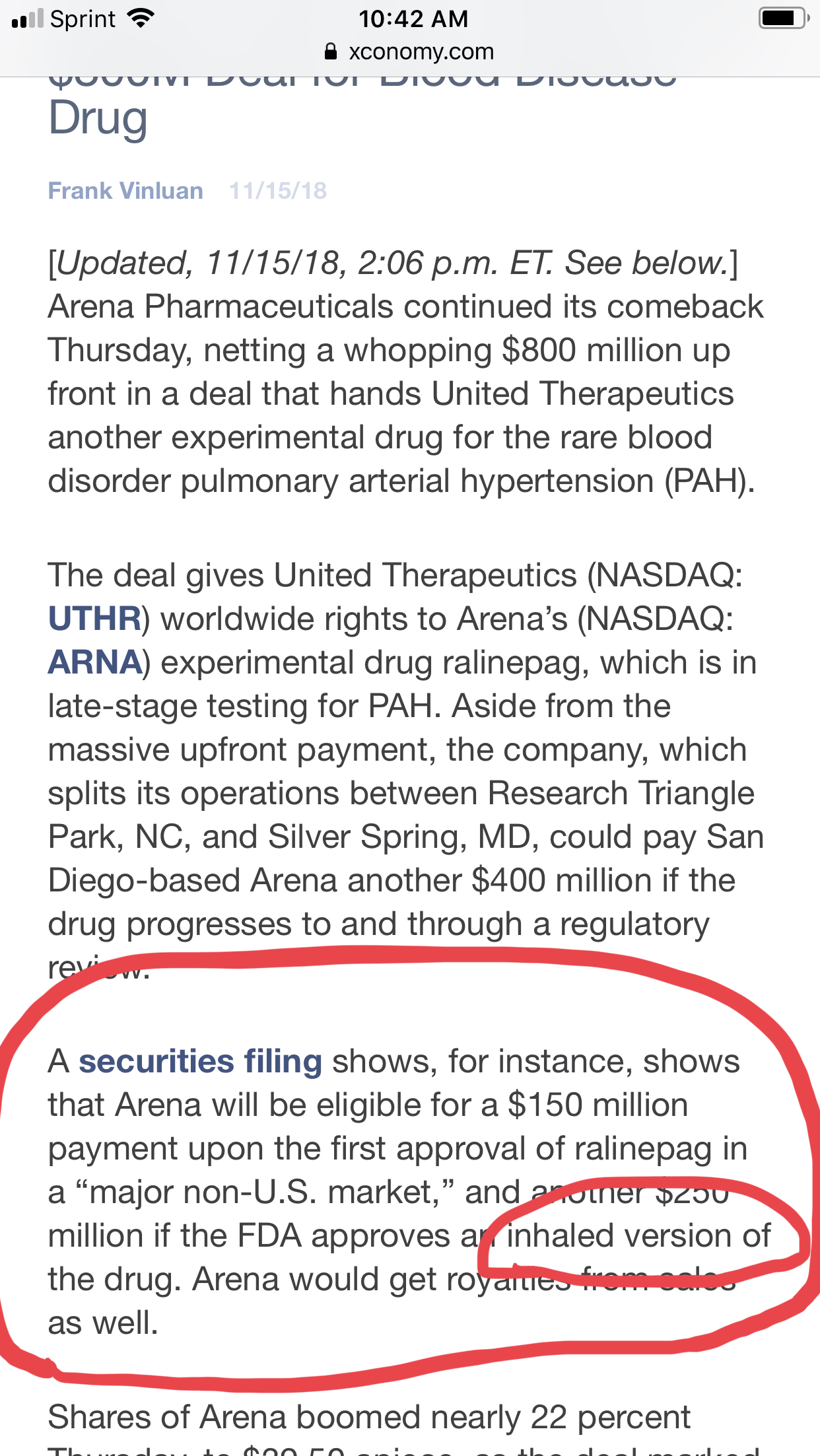 |
|
|
|
Post by hammer on Nov 16, 2018 10:57:06 GMT -5
"The parties also entered into a research agreement for the conduct of research by MannKind on behalf of United Therapeutics for products outside the scope of the licensing and collaboration agreement. MannKind will receive an immediate payment of $10 million in consideration for its performance under the research agreement."
I think MNKD is tasked with making Ralinepag inhalable.
So Arena get 250mil if mannkind is successful. What would Mannkind receive?
|
|
|
|
Post by mnkdfann on Nov 16, 2018 11:11:06 GMT -5
"The parties also entered into a research agreement for the conduct of research by MannKind on behalf of United Therapeutics for products outside the scope of the licensing and collaboration agreement. MannKind will receive an immediate payment of $10 million in consideration for its performance under the research agreement." I think MNKD is tasked with making Ralinepag inhalable.So Arena get 250mil if mannkind is successful. What would Mannkind receive? Spencer addresses the point you raised in the comments to his latest article. The following text is quoted from there: Ralinepag is an extended release once per day pill. Logic indicates that putting it on Technosphere is not necessary. Technosphere is best at quick release. Thus:
1. Why take puffs throughout the day when you can simply take 1 pill?
2. Why would United have any desire to pay Mannkind $40 million plus royalties to put Ralinepag on technosphere? |
|
|
|
Post by uvula on Nov 16, 2018 11:14:07 GMT -5
So Arena get 250mil if mannkind is successful. What would Mannkind receive? By that time Mannkind will already be a division of United Therapeutics. |
|
|
|
Post by Clement on Nov 16, 2018 11:22:27 GMT -5
I wonder if Danbury is already working on DPI ralinepag. Thanks for your post, Harry!
(DPI = Dry Powder Inhaled)
|
|
|
|
Post by harryx1 on Nov 16, 2018 11:27:56 GMT -5
IMO, it's not about the money for Martine, it's about having all possible options for people with PAH. I think UT would happily take 10-20% of the revs for any drug that would greatly improve a person's life with PAH.  |
|
|
|
Post by awesomo on Nov 16, 2018 12:09:14 GMT -5
Yeah, she seems like a reputable leader, but UTHR is still a public company that needs to satisfy investors and Wall Street. And when you pay 20x for something (45M vs 800M upfront), you better damn well prioritize that or there will be tons are questions from said investors.
|
|
|
|
Post by sportsrancho on Nov 16, 2018 12:39:42 GMT -5
|
|
|
|
Post by mango on Nov 16, 2018 13:31:09 GMT -5
"The parties also entered into a research agreement for the conduct of research by MannKind on behalf of United Therapeutics for products outside the scope of the licensing and collaboration agreement. MannKind will receive an immediate payment of $10 million in consideration for its performance under the research agreement." I think MNKD is tasked with making Ralinepag inhalable.So Arena get 250mil if mannkind is successful. What would Mannkind receive? Spencer addresses the point you raised in the comments to his latest article. The following text is quoted from there: Ralinepag is an extended release once per day pill. Logic indicates that putting it on Technosphere is not necessary. Technosphere is best at quick release. Thus:
1. Why take puffs throughout the day when you can simply take 1 pill?
2. Why would United have any desire to pay Mannkind $40 million plus royalties to put Ralinepag on technosphere?Ralinepag is a selective prostacyclin receptor agonist. Treprostinil is a prostaglandin I2 analog. 1. Why take puffs throughout the day when you can simply take 1 pill? Treprostinil Technosphere is ancipated to allow people with PAH to achieve at or beyond the current recommended total daily dose of treprostinil (216mcg) in a single inhalation, as already evidenced by Mike Castagna's slides on Science Day. TreT was tolerated in healthy volunteers up through 150mcg. For comparison, Arena's results from their single-dose escalation study involving healthy volunteers showed Ralinepag was tolerated up to 0.1 mg. That's 100mcg. TreT was tolerated up through 150mcg, far exceeding Ralinepag's tolerability already in healthy volunteers. Dose escalation for Ralinepag was discontinued at 0.2 mg due to treatment-emergent AEs (vomiting, headache, and nausea). There is no anticipation, nor evidence indicating, that people with PAH will potentially require taking "puffs throughout the day" with TreT, as Spencer so incorrectly stated. 2. Why would United have any desire to pay Mannkind $40 million plus royalties to put Ralinepag on technosphere? Ralinepag is a selective IP agonist and is different from the analog Treprostinil. If Ralinepag demonstrates promising therapeutic potential, beyond what Treprostinil can provide, or would be a great combo, etc...but shows drawbacks and limitations mainly due to its ROA &/or formulation, then United Therapeutics could get MannKind to formulate it onto Technosphere to overcome those barriers. |
|
|
|
Post by traderdennis on Nov 16, 2018 13:36:45 GMT -5
Yeah, she seems like a reputable leader, but UTHR is still a public company that needs to satisfy investors and Wall Street. And when you pay 20x for something (45M vs 800M upfront), you better damn well prioritize that or there will be tons are questions from said investors. Plus the 250MM for MNKD is a poison pill for ARNA. Gives more incentive to make sure ARNA's solution is primary. |
|
|
|
Post by liane on Nov 16, 2018 14:25:17 GMT -5
Ralinepag is a selective prostacyclin receptor agonist. Treprostinil is a prostaglandin I2 analog. 1. Why take puffs throughout the day when you can simply take 1 pill? Treprostinil Technosphere is ancipated to allow people with PAH to achieve at or beyond the current recommended total daily dose of treprostinil (216mcg) in a single inhalation, as already evidenced by Mike Castagna's slides on Science Day. TreT was tolerated in healthy volunteers up through 150mcg. For comparison, Arena's results from their single-dose escalation study involving healthy volunteers showed Ralinepag was tolerated up to 0.1 mg. That's 100mcg. TreT was tolerated up through 150mcg, far exceeding Ralinepag's tolerability already in healthy volunteers. Dose escalation for Ralinepag was discontinued at 0.2 mg due to treatment-emergent AEs (vomiting, headache, and nausea). There is no anticipation, nor evidence indicating, that people with PAH will potentially require taking "puffs throughout the day" with TreT, as Spencer so incorrectly stated. 2. Why would United have any desire to pay Mannkind $40 million plus royalties to put Ralinepag on technosphere? Ralinepag is a selective IP agonist and is different from the analog Treprostinil. If Ralinepag demonstrates promising therapeutic potential, beyond what Treprostinil can provide, or would be a great combo, etc...but shows drawbacks and limitations mainly due to its ROA &/or formulation, then United Therapeutics could get MannKind to formulate it onto Technosphere to overcome those barriers. You cannot compare mcg TreT to mcg Ralinepag. 2 different drugs, different molecular wts, different effects. |
|
|
|
Post by lakers on Nov 16, 2018 14:31:31 GMT -5
invest.arenapharm.com/node/19076/html“ including covenants to cooperate in seeking regulatory approvals, as well as Arena’s agreement not to compete, during the period in which royalties are payable (or during the five-year period following the closing if Arena is subject to a change of control transaction) in the development of a prostacyclin to treat pulmonary hypertension (“PH”). The Agreement does not contain a covenant obligating United Therapeutics to use any particular efforts to develop or commercialize any Product. Arena has also agreed to grant United Therapeutics, for a period of six years following the closing, certain rights to negotiate for potential access to future compounds developed by Arena for the treatment, prevention or amelioration of PH.” UTHR really corners the PAH market. Wonder if they will pass HSR Act. “Under the terms of the License Agreement, Arena will receive an upfront payment of $800 million at the closing of the transaction, Arena will be eligible to receive a payment of $150 million upon first marketing approval of ralinepag in a major non-U.S. market, and Arena will be eligible to receive a payment of $250 million upon U.S. marketing approval of an inhaled formulation of ralinepag. In addition, Arena will be entitled to receive low double-digit, tiered royalties on net sales of Products, subject to certain adjustments for third party license payments. In addition, if the closing of the transaction occurs after December 1, 2018, United Therapeutics will pay Arena, at closing, an additional amount based on the number of months lapsed between December 1, 2018 and closing and the achievement of certain agreed upon clinical goals relating to the Compound. Arena expects a significant portion of the taxable gain that would otherwise be triggered by the upfront payment will be offset by Arena’s net operating losses.” If there is no merit to an inhaled formulation of ralinepag and its U.S. marketing approval, why is that part of the deal at all, let alone the huge $250M Incentive in U.S. alone for such a brain dead product? Maybe ARNA, UTHR, MNKD are brain dead together as SO thought. Think Different!
Waiting for AbbVie to copy a chapter from UTHR for the $22B (2018 forecast) DPI Humira to protect its cash cow from the generics onslaughts.
A new inhaled version would protect and extend their patent on Humira, and to ward off Multiple biosimilars and Lilly’s NCE. Eli Lilly’s Arthritis Drug Approved at Lower Dose with Boxed Warning Frank Vinluan 6/1/18 www.xconomy.com/indiana/2018/06/01/eli-lillys-arthritis-drug-approved-at-lower-dose-with-boxed-warning/Indianapolis-based Lilly said baricitinib will launch by the end of the month, priced at a more than 60 percent discount to blockbuster AbbVie (NYSE: ABBV) RA drug adalimumab (Humira). Adalimumab’s annual cost tops $40,000, according to calculations by the Institute for Clinical and Economic Review, a non-profit group and drug price watchdog. Because the FDA approval extends only to those patients who have not adequately responded to TNF blocking drugs such as adalimumab, it will be harder for Lilly to take market share from the AbbVie drug, which generated more than $18 billion in 2017 revenue. Baricitinib was originally developed by Wilmington, DE-based Incyte (NASDAQ: INCY). In 2009, Lilly licensed rights to develop the drug for inflammatory and autoimmune diseases. Following baricitinib’s approval, Incyte is now eligible for a $100 million milestone payment from its partner. Baricitinib was approved in the EU in February 2017 and in Japan in July 2017. investor.lilly.com/news-releases/news-release-details/lilly-and-incyte-announce-collaboration-development-and Under the terms of the agreement, Lilly will receive worldwide rights to develop and commercialize INCB28050 as an oral treatment for all inflammatory conditions. In exchange for these rights, Incyte will receive an initial payment of $90 million and is eligible for up to $665 million in additional potential development, regulatory, and commercialization milestones, as well as tiered, double-digit royalty payments on future global sales with rates ranging up to twenty percent if a product is successfully commercialized. Read more: mnkd.proboards.com/thread/10427/next-potential-apps?page=12#ixzz5X3CE0nf3Dr. Castagna, was Executive Director of Bristol-Myers Squibb’s Immunology franchise, where he served as co-lead to relaunch Orencia IV and launch Orencia SC, both rheumatoid arthritis drugs.
|
|
|
|
Post by centralcoastinvestor on Nov 16, 2018 14:36:03 GMT -5
Spencer addresses the point you raised in the comments to his latest article. The following text is quoted from there: Ralinepag is an extended release once per day pill. Logic indicates that putting it on Technosphere is not necessary. Technosphere is best at quick release. Thus:
1. Why take puffs throughout the day when you can simply take 1 pill?
2. Why would United have any desire to pay Mannkind $40 million plus royalties to put Ralinepag on technosphere?Ralinepag is a selective prostacyclin receptor agonist. Treprostinil is a prostaglandin I2 analog. 1. Why take puffs throughout the day when you can simply take 1 pill? Treprostinil Technosphere is ancipated to allow people with PAH to achieve at or beyond the current recommended total daily dose of treprostinil (216mcg) in a single inhalation, as already evidenced by Mike Castagna's slides on Science Day. TreT was tolerated in healthy volunteers up through 150mcg. For comparison, Arena's results from their single-dose escalation study involving healthy volunteers showed Ralinepag was tolerated up to 0.1 mg. That's 100mcg. TreT was tolerated up through 150mcg, far exceeding Ralinepag's tolerability already in healthy volunteers. Dose escalation for Ralinepag was discontinued at 0.2 mg due to treatment-emergent AEs (vomiting, headache, and nausea). There is no anticipation, nor evidence indicating, that people with PAH will potentially require taking "puffs throughout the day" with TreT, as Spencer so incorrectly stated. 2. Why would United have any desire to pay Mannkind $40 million plus royalties to put Ralinepag on technosphere? Ralinepag is a selective IP agonist and is different from the analog Treprostinil. If Ralinepag demonstrates promising therapeutic potential, beyond what Treprostinil can provide, or would be a great combo, etc...but shows drawbacks and limitations mainly due to its ROA &/or formulation, then United Therapeutics could get MannKind to formulate it onto Technosphere to overcome those barriers. Well stated. Thanks for posting this. |
|