|
|
Post by peppy on Jun 2, 2021 1:19:08 GMT -5
Yep. I listened to UTHR at the Jeffries Conference today. James Edmund CFO gave a good summary and they still expect approval in December. But we have heard that before. Specifically, he said "pending acceptance and approval" when he talked about Tyvaso DPI NDA submission. That means to me that FDA has not yet "accepted" the submission, which I have been hoping for any day now. reminds me of a song: "Any day now, any day now, I shall be released." written by Bob Dylan 1967 Yeah, I know. I'm old if I remember it. |
|
|
|
Post by mytakeonit on Jun 2, 2021 1:31:03 GMT -5
Wow !!! I don't remember it ... so I'm not OLD !!! Seems like a lot of remembrance in that clip.
But, that's mytakeonit
|
|
|
|
Post by peppy on Jun 2, 2021 1:54:26 GMT -5
Not to be confused with.....  The Band, And The Staples - The Weight (The Last Waltz) |
|
|
|
Post by sportsrancho on Jun 2, 2021 7:35:50 GMT -5
I think it’s the delay in the FDA OK for the submission. Not sure... but the stocks gonna bleed out with no news. Very annoying. Yep. I listened to UTHR at the Jeffries Conference today. James Edmund CFO gave a good summary and they still expect approval in December. But we have heard that before. Specifically, he said "pending acceptance and approval" when he talked about Tyvaso DPI NDA submission. That means to me that FDA has not yet "accepted" the submission, which I have been hoping for any day now. reminds me of a song: "Any day now, any day now, I shall be released." And here’s the reason we had a bad day yesterday... finance.yahoo.com/news/liquidia-announces-fda-acceptance-drug-110000563.html |
|
|
|
Post by Clement on Jun 2, 2021 7:44:58 GMT -5
Yep. I listened to UTHR at the Jeffries Conference today. James Edmund CFO gave a good summary and they still expect approval in December. But we have heard that before. Specifically, he said "pending acceptance and approval" when he talked about Tyvaso DPI NDA submission. That means to me that FDA has not yet "accepted" the submission, which I have been hoping for any day now. reminds me of a song: "Any day now, any day now, I shall be released." And here’s the reason we had a bad day yesterday... finance.yahoo.com/news/liquidia-announces-fda-acceptance-drug-110000563.htmlFrom the PR: "If the FDA determines, following its substantive review of the NDA, that all requirements for approval have been met, the FDA may issue tentative approval on a timeline generally informed by the PDUFA goal date. Any final FDA approval of the NDA for LIQ861 would be subject to the resolution of the pending Hatch-Waxman litigation commenced by United Therapeutics, and also subject to the FDA’s consideration of developments that may have occurred since the time of the tentative approval. The FDA may not issue a final approval until the expiration of a 30-month regulatory stay in October 2022 or an earlier judgment unfavorable to United Therapeutics by the court. A new drug product may not be marketed until the date of final approval which is confirmed by the FDA at the time of final submission." |
|
|
|
Post by uvula on Jun 2, 2021 7:59:39 GMT -5
Why did so many people know about the liquidia news before we did? Was anything public or was this illegal insider trading?
|
|
|
|
Post by peppy on Jun 2, 2021 8:06:47 GMT -5
I see COPD. 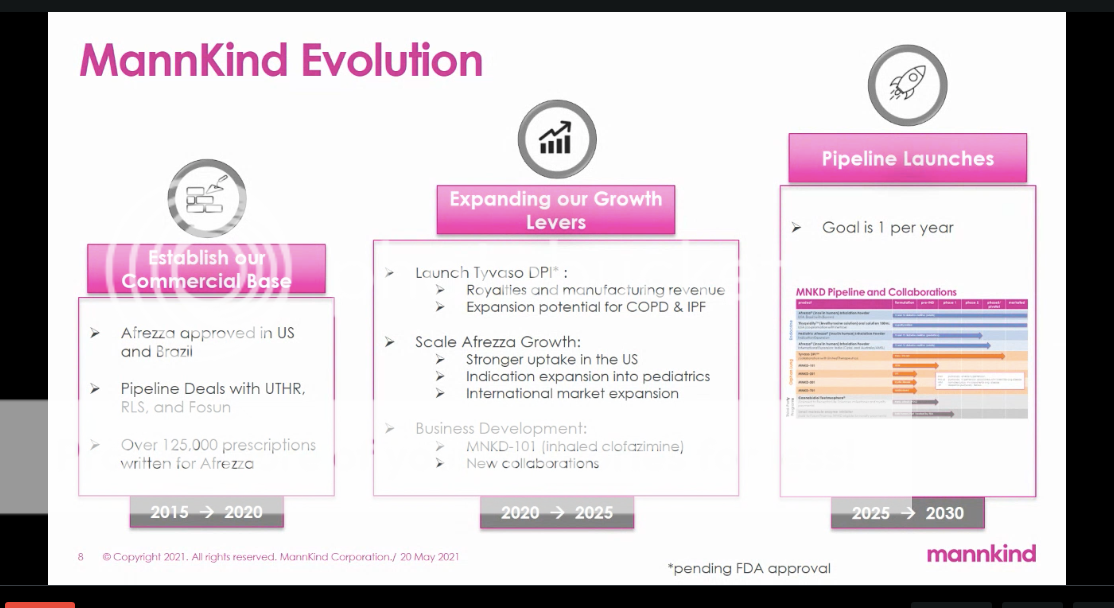 |
|
|
|
Post by Clement on Jun 2, 2021 8:11:47 GMT -5
Yep. I listened to UTHR at the Jeffries Conference today. James Edmund CFO gave a good summary and they still expect approval in December. But we have heard that before. Specifically, he said "pending acceptance and approval" when he talked about Tyvaso DPI NDA submission. That means to me that FDA has not yet "accepted" the submission, which I have been hoping for any day now. reminds me of a song: "Any day now, any day now, I shall be released." And here’s the reason we had a bad day yesterday... finance.yahoo.com/news/liquidia-announces-fda-acceptance-drug-110000563.htmlAlso from the PR: "Liquidia Corporation (NASDAQ: LQDA) announced today that the U.S. Food and Drug Administration (FDA) has accepted its New Drug Application (NDA) resubmission for LIQ861 (treprostinil) inhalation powder to treat pulmonary arterial hypertension (PAH)." UTHR at the Jeffries Conference yesterday said that the PAH space already has multiple therapies available and is somewhat crowded already. Where UTHR is planning for growth is in the additional indications - the first of which is PH-ILD. PH-ILD has no currently approved therapies, so it's a good target for growth. When Tyvaso DPI is approved hopefully in December, it is expected to be approved for both PAH and PH-ILD. The LQDA press release does not mention any additional indications other than PAH. |
|
|
|
Post by Clement on Jun 2, 2021 8:43:39 GMT -5
|
|
|
|
Post by sportsrancho on Jun 2, 2021 8:54:12 GMT -5
Why did so many people know about the liquidia news before we did? Was anything public or was this illegal insider trading? I was told by an attorney:-) The one you know... that it’s the attorneys that usually leak it. |
|
|
|
Post by peppy on Jun 2, 2021 9:30:45 GMT -5
|
|
|
|
Post by uvula on Jun 2, 2021 9:58:31 GMT -5
Since bad news for UTHR leads to a larger percentage drop in MNKD than in UTHR, it is pretty clear that everyone has given up on Afrezza ever being a money making drug. (Even though most agree that Afrezza is a great insulin option for some people.)
|
|
|
|
Post by mymann on Jun 2, 2021 10:08:09 GMT -5
There's the volume we were looking for. Going the wrong way.
|
|
|
|
Post by hellodolly on Jun 2, 2021 10:14:20 GMT -5
Love these sweet swings!
|
|
|
|
Post by joeypotsandpans on Jun 2, 2021 10:23:57 GMT -5
Since bad news for UTHR leads to a larger percentage drop in MNKD than in UTHR, it is pretty clear that everyone has given up on Afrezza ever being a money making drug. (Even though most agree that Afrezza is a great insulin option for some people.) Uvula, I have not given up as you alluded to in your prior post in the scripts thread, rather quite the contrary. You misinterpreted what I posted as I was saying that MNKD was not as dependent on Afrezza as it once was. My emphasis was on the value of Afrezza validating technosphere as a progressive way of treating future diseases. I never said that Afrezza would never be profitable, that was YOUR "twist" on my post. Regarding the difference in percentage loss between UTHR and MNKD sp, UTHR has a vastly different valuation and price multiple vs. earnings than MNKD so it's like comparing apples and oranges IMO. I respectfully disagree with the generalization that "everyone" has given up on Afrezza ever being a money making drug. 😉 |
|