|
|
Post by EveningOfTheDay on Nov 30, 2015 23:54:03 GMT -5
I feel like bad or good news the market punishes MannKind. I'm wondering if Sanofi is ultimately not the answer to unlock MannKind's true hidden value. If Al Mann were smart he would have already been seeking multiple partnerships by now. I don't know which way to guess this story will go. I am tired of guessing. I just want to see facts. I have been guessing wrong for almost a year now. Time frame for my ultimate guess, two years from approval, have not come around yet, but as it approaches without any perceivable improvement is forcing me to really take a hard look at the situation. With the sp in the doldrums and no good news to lift the hope, my confidence has completely vanished. Yet I still carry that little nagging voice that tells me I could not possibly have been that wrong. I wish it would go away because it certainly complicates the decision making. |
|
|
|
Post by dreamboatcruise on Dec 1, 2015 0:37:19 GMT -5
mnholdem... sorry, I did forget the part about diabetic ketoacidosis. I guess I have been discounting that as being a minor issue with regard to prescribing. This isn't going to change anything with regard to claims about A1c or risk of hypos, since those things aren't even measured, but if scrips improve even slightly because of removal of ketoacidosis warning that will be good.
|
|
|
|
Post by lakers on Dec 1, 2015 1:38:14 GMT -5
Just as Toujeo failed to claim lower hypo incidents in U.S., but in EU, Sanofi might be able to obtain a better A1c label overseas based on Affinity 2 Study. The TI arm demonstrated a significantly greater reduction in HbA1c at week 24 compared with the TP arm, with a difference of −0.40 percentage points (P < 0.001). A larger percentage of patients in the TI group achieved the desired HbA1c goal of less than 7% compared with the TP group (37.7% versus 19.0%, respectively; P = 0.0005). The mean change in FPG levels was not significantly different between the two groups.24 During the 24-week study period, the patients receiving TI experienced a mean 0.49-kg weight gain, whereas those in the placebo group lost an average of 1.13 kg; the between-group treatment difference was 1.62 kg (P < 0.0001).22 Read more: mnkd.proboards.com/thread/4198/forecast-great-afrezza-article-change#ixzz3t348OdRSIn 2016, $30M for EU Approval. $25M for qualifying Sny's insulin. In 2017, potentially 12 countries of $20.84M annual rev each = $250M qualifying for the first $250M milestone. This also enables Mnkd to recognize deferred rev, and upfront payment. Looking forward, RBC identified three potential catalysts for the stock: 1. European Union filing plans in 2016 in which MannKind will count on Sanofi SA (NYSE: SNY) for an MAA filing where visibility on approval would be a positive. 2. Afrezza post-approval study details as timelines have implications for label expansion. 3. Launch trajectory over the next 12 months. |
|
|
|
Post by thoth on Dec 1, 2015 14:20:23 GMT -5
The fact that the studies were ended early is a very good sign. Problematic clinical studies usually end up being delayed because the researchers are frantically trying to gather more data or figure out new ways to manipulate existing data to get a positive result. It sounds like in both of these cases that everything went extremely well.
|
|
|
|
Post by EveningOfTheDay on Dec 1, 2015 14:29:04 GMT -5
The fact that the studies were ended early is a very good sign. Problematic clinical studies usually end up being delayed because the researchers are frantically trying to gather more data or figure out new ways to manipulate existing data to get a positive result. It sounds like in both of these cases that everything went extremely well. I fully agree with your assessment, which incidentally would also concur with what mostly all the users that have made their experience public are saying in avery short period of time after initiating treatment with Afrezza. I sure hope Sanofi is able to maximize the positive results to boost Afrezza sales. |
|
|
|
Post by dreamboatcruise on Dec 1, 2015 14:33:01 GMT -5
lakers... yes, it would be hugely positive if EU authorities look at Affinity trials in a different light and have label suggesting better clinical outcomes. The question would be likelihood of this. Obviously the new trials have not added to the data regarding A1c.
|
|
|
|
Post by mango on Apr 20, 2017 20:22:29 GMT -5
Looks like they were updated again. PK and PD Within-Subject Variability of a Single Dose of Afrezza Inhaled Technosphere Insulin in Patients With Diabetes Mellitus Type 1 (T1DM)This study has been completed. Sponsor: Mannkind Corporation Information provided by (Responsible Party): Mannkind Corporation • First received: June 25, 2015 • Last updated: March 13, 2017• Last verified: March 2017 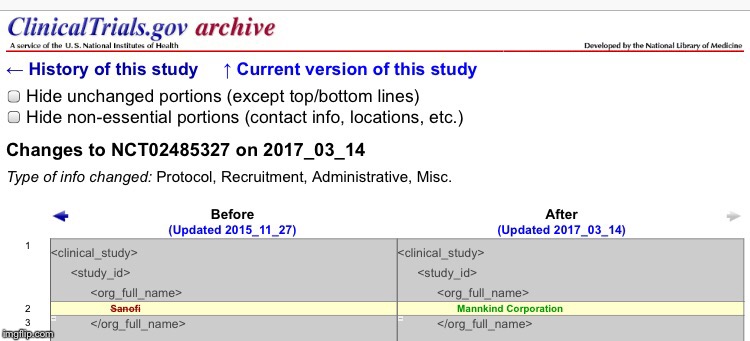 Responsible Party: Mannkind Corporation ClinicalTrials.gov Identifier: NCT02485327 History of Changes Other Study ID Numbers: PDY14329 2015-001008-74 ( EudraCT Number ) U1111-1168-4442 ( Other Identifier: UTN ) Study First Received: June 25, 2015 Last Updated: March 13, 2017 clinicaltrials.gov/ct2/show/NCT02485327?term=MannKind&rank=26&submit_fld_opt=Single Dose Clamp Study to Evaluate Concentration-time Profile and Metabolic Activity of 3 Dose Levels of Afrezza and 3 Dose Levels of Insulin Lispro in Patients With Type 1 Diabetes MellitusThis study has been completed. Sponsor: Mannkind Corporation Information provided by (Responsible Party): Mannkind Corporation • First received: June 10, 2015 • Last updated: April 12, 2017• Last verified: April 2017 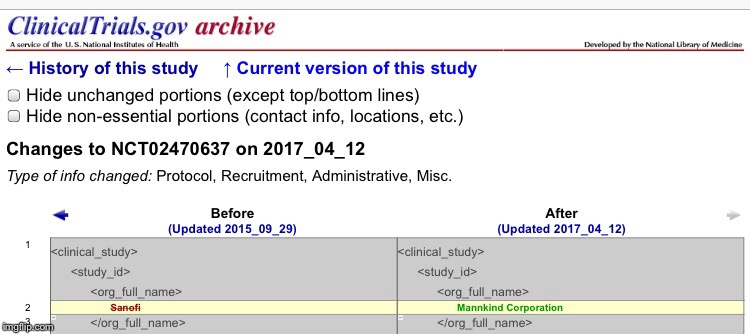 Responsible Party: Mannkind Corporation ClinicalTrials.gov Identifier: NCT02470637 History of Changes Other Study ID Numbers: PDY14324 2015-000231-33 ( EudraCT Number ) U1111-1166-5431 ( Other Identifier: UTN ) Study First Received: June 10, 2015 Last Updated: April 12, 2017 clinicaltrials.gov/ct2/show/NCT02470637?term=MannKind&rank=27&submit_fld_opt= |
|
|
|
Post by mango on Apr 20, 2017 21:12:25 GMT -5
I thought this was kind of interesting and probably means nothing at all. I just thought it was strange.  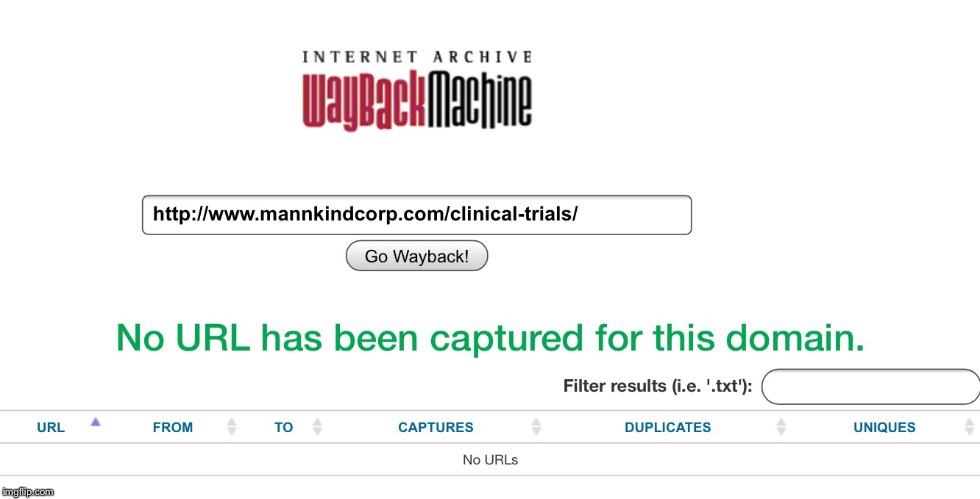 I realize they left off the 'al' in Costal Clinical Research Inc, but that isn't important. They have been a clinical trial site for MannKind before, but why would MannKind have this page created with only this one independent multi-specialty research site listed. Why would MannKind have this page created at all? www.mannkindcorp.com/clinical-trials/www.coastalclinicalresearch.com/clinical-trial-research.html |
|
Deleted
Deleted Member
Posts: 0
|
Post by Deleted on Apr 20, 2017 23:03:27 GMT -5
I think what is even more interesting is Lakers on the money as usually earlier on in the thread
|
|