Deleted
Deleted Member
Posts: 0
|
Post by Deleted on Jan 2, 2017 20:45:35 GMT -5
Is Mannkind still working on the protocol with FDA?
How much time does this protocol take to finalize and start recruiting?
1 yr anniversary of Sanofi dumping Afrezza is coming soon.
its one thing Sanofi dragging their feet, but Mannkind?
|
|
|
|
Post by mnholdem on Jan 2, 2017 21:00:13 GMT -5
Dr. Raymond Urbanski - Chief Medical Officer, MannKind Corporation
Excerpt from presentation at 3Q16 Earnings Call, November 9, 2016:
Next slide, now looking at our clinical development program for Afrezza, we recently met with several of our pediatric steering committee members and are working through some potential protocol changes for the PK portion of this program. In addition, we have identified key partners which we have announced previous for our pediatric program and they are going to replace and already are supporting our activities.
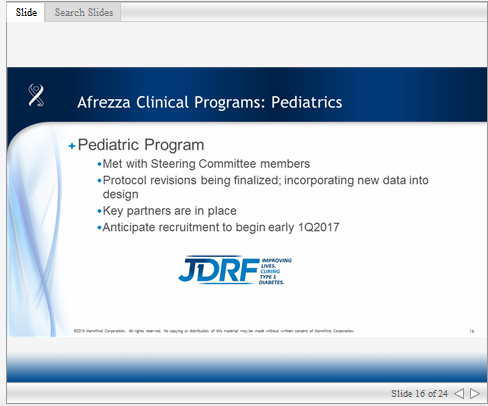
Next slide, the subsequent pediatric Phase 3 trial which we will file for pediatric indication, we will incorporate evidence not only from the PK study that we have conducted previously but as well as data from recent publications. These publications, as you can see on this slide, include dosing simulation work and the Afrezza with the artificial pancreas. The use of Afrezza with the artificial pancreas is impressive and promising and an area that we are actively investigating.
When you take all of this data around the dosing and titration of Afrezza it helps to inform us as to the proposed pediatric protocol changes that we will need in the Phase 3 trial, which we believe will substantially improve study recruitment and retention and thereby expediting our filing dates. We are planning for study execution to begin in the first quarter of 2017.
|
|
|
|
Post by centralcoastinvestor on Jan 3, 2017 11:43:49 GMT -5
I was wondering how news worthy the official launch of the new pediatric study will be. When is the last time a new type of insulin treatment was being studied for kids? This could be an opportunity for Afrezza visibility with JDRF being a partner on this effort.
|
|
|
|
Post by careful2invest on Jan 3, 2017 11:54:14 GMT -5
I was wondering how news worthy the official launch of the new pediatric study will be. When is the last time a new type of insulin treatment was being studied for kids? This could be an opportunity for Afrezza visibility with JDRF being a partner on this effort. Exactly, It's events like this that MNKD can organically put together a story that is newsworthy to get the word out about AFREZZA. Hopefully, MNKD seizes this opportunity, instead of yet another missed opportunity. We're all waiting/watching. Show us something fellas!! GLTA TRUE LONGS! |
|
|
|
Post by nylefty on Jan 3, 2017 12:04:28 GMT -5
I was wondering how news worthy the official launch of the new pediatric study will be. When is the last time a new type of insulin treatment was being studied for kids? This could be an opportunity for Afrezza visibility with JDRF being a partner on this effort. Exactly, It's events like this that MNKD can organically put together a story that is newsworthy to get the word out about AFREZZA. Hopefully, MNKD seizes this opportunity, instead of yet another missed opportunity. We're all waiting/watching. Show us something fellas!! GLTA TRUE LONGS! What were all the "missed" opportunities you refer to? |
|
|
|
Post by careful2invest on Jan 3, 2017 12:13:31 GMT -5
Exactly, It's events like this that MNKD can organically put together a story that is newsworthy to get the word out about AFREZZA. Hopefully, MNKD seizes this opportunity, instead of yet another missed opportunity. We're all waiting/watching. Show us something fellas!! GLTA TRUE LONGS! What were all the "missed" opportunities you refer to? Since the announcement of the FDA approval of AFREZZA, to the partnering with Sanofi (although turned out to be a bust) To joining with JDRF and other opportunities as well. None of those events were expanded on at all to gain notoriety. The silence has been much more prevelant than the announcements. And here we are, entering our third year after the FDA approval, and so many have not even heard of AFREZZA. IMHO, It does not make good business sense. GLTA TRUE LONGS! |
|
Deleted
Deleted Member
Posts: 0
|
Post by Deleted on Jan 3, 2017 12:19:10 GMT -5
Since the announcement of the FDA approval of AFREZZA, to the partnering with Sanofi (although turned out to be a bust) To joining with JDRF and other opportunities as well. None of those events were expanded on at all to gain notoriety. The silence has been much more prevelant than the announcements. And here we are, entering our third year after the FDA approval, and so many have not even heard of AFREZZA. IMHO, It does not make good business sense. GLTA TRUE LONGS! you should probably wish GLTA TRUE MANAGEMENT hehe |
|
|
|
Post by wildthing on Jan 3, 2017 12:34:46 GMT -5
Dr. Raymond Urbanski - Chief Medical Officer, MannKind Corporation
Excerpt from presentation at 3Q16 Earnings Call, November 9, 2016:
Next slide, now looking at our clinical development program for Afrezza, we recently met with several of our pediatric steering committee members and are working through some potential protocol changes for the PK portion of this program. In addition, we have identified key partners which we have announced previous for our pediatric program and they are going to replace and already are supporting our activities.

Next slide, the subsequent pediatric Phase 3 trial which we will file for pediatric indication, we will incorporate evidence not only from the PK study that we have conducted previously but as well as data from recent publications. These publications, as you can see on this slide, include dosing simulation work and the Afrezza with the artificial pancreas. The use of Afrezza with the artificial pancreas is impressive and promising and an area that we are actively investigating.

When you take all of this data around the dosing and titration of Afrezza it helps to inform us as to the proposed pediatric protocol changes that we will need in the Phase 3 trial, which we believe will substantially improve study recruitment and retention and thereby expediting our filing dates. We are planning for study execution to begin in the first quarter of 2017.
What were the proposed protocol changes? |
|
|
|
Post by mnholdem on Jan 3, 2017 12:45:44 GMT -5
Ray wasn't specific, but the data from the study, "Improving Efficacy of Inhaled Technosphere Insulin (Afrezza) by Postmeal Dosing..." may be what he was referring to. MannKind must establish protocols that will demonstrate the very best efficacy possible for these pediatric trials.
Non-inferior is not good enough, IMHO. MannKind must have the objective of demonstrating via these Phase IV trials that Afrezza is clearly a superior prandial insulin for pediatric use. I'm also a bit excited about his mention of using Afrezza with artificial pancreas systems. |
|
|
|
Post by nylefty on Jan 3, 2017 12:55:10 GMT -5
What were all the "missed" opportunities you refer to? Since the announcement of the FDA approval of AFREZZA, to the partnering with Sanofi (although turned out to be a bust) To joining with JDRF and other opportunities as well. None of those events were expanded on at all to gain notoriety. The silence has been much more prevelant than the announcements. And here we are, entering our third year after the FDA approval, and so many have not even heard of AFREZZA. IMHO, It does not make good business sense. GLTA TRUE LONGS! And just how should have those events been "expanded" to "gain notoriety?" I spent many years in radio and TV news and haven't seen any events in MannKind's history that merited much news coverage. Afrezza isn't the first inhaled insulin and therefore is not "new," period. Yes, we know that it's better than an earlier inhaled insulin, but the (flawed) studies haven't really shown that (yet). So far, the story is that a second attempt to market an inhaled insulin hasn't been successful so far. Not much of a story (yet). However, I still remain bullish on MNKD. |
|
|
|
Post by centralcoastinvestor on Jan 3, 2017 13:16:38 GMT -5
Ray wasn't specific, but the data from the study, "Improving Efficacy of Inhaled Technosphere Insulin (Afrezza) by Postmeal Dosing..." may be what he was referring to. MannKind must establish protocols that will demonstrate the very best efficacy possible for these pediatric trials.
Non-inferior is not good enough, IMHO. MannKind must have the objective of demonstrating via these Phase IV trials that Afrezza is clearly a superior prandial insulin for pediatric use. I'm also a bit excited about his mention of using Afrezza with artificial pancreas systems. mnholdem, your post triggered an exciting possibility that had not occurred to me before although it should have. Whatever results that are determined in the pediatric study can easily be used to improve the label for adults. If they approve it for kids, adult use is a no brainer. So if MannKind, JDRF and the FDA can established protocols that showcase Afrezza in its highest and best use i.e. lower hypoglycemia events, markedly improved A1c, improved glucose control, we won't need another study to improve the overall label. JMHO |
|
|
|
Post by wildthing on Jan 3, 2017 13:26:49 GMT -5
Ray wasn't specific, but the data from the study, "Improving Efficacy of Inhaled Technosphere Insulin (Afrezza) by Postmeal Dosing..." may be what he was referring to. MannKind must establish protocols that will demonstrate the very best efficacy possible for these pediatric trials.
Non-inferior is not good enough, IMHO. MannKind must have the objective of demonstrating via these Phase IV trials that Afrezza is clearly a superior prandial insulin for pediatric use. I'm also a bit excited about his mention of using Afrezza with artificial pancreas systems. mnholdem, your post triggered an exciting possibility that had not occurred to me before although it should have. Whatever results that are determined in the pediatric study can easily be used to improve the label for adults. If they approve it for kids, adult use is a no brainer. So if MannKind, JDRF and the FDA can established protocols that showcase Afrezza in its highest and best use i.e. lower hypoglycemia events, markedly improved A1c, improved glucose control, we won't need another study to improve the overall label. JMHO But the pediatrics trial is just a phase 1 study, isn't it? That's just to determine whether it's safe for further studies to determine effects on A1c, etc. |
|
|
|
Post by agedhippie on Jan 3, 2017 13:36:33 GMT -5
mnholdem, your post triggered an exciting possibility that had not occurred to me before although it should have. Whatever results that are determined in the pediatric study can easily be used to improve the label for adults. If they approve it for kids, adult use is a no brainer. So if MannKind, JDRF and the FDA can established protocols that showcase Afrezza in its highest and best use i.e. lower hypoglycemia events, markedly improved A1c, improved glucose control, we won't need another study to improve the overall label. JMHO I would be surprised if you could change the adult label on the basis of the pediatric trial. The trial data is for a pediatric population, not an adult population. For a start children tend to be far more insulin sensitive than adults because of body weight. I think changing the adult label will require further trials to demonstrate the improvement in an adult population. I would happily take a superiority finding for pediatrics though - it's a start. |
|
|
|
Post by careful2invest on Jan 3, 2017 15:24:01 GMT -5
Ray wasn't specific, but the data from the study, "Improving Efficacy of Inhaled Technosphere Insulin (Afrezza) by Postmeal Dosing..." may be what he was referring to. MannKind must establish protocols that will demonstrate the very best efficacy possible for these pediatric trials.
Non-inferior is not good enough, IMHO. MannKind must have the objective of demonstrating via these Phase IV trials that Afrezza is clearly a superior prandial insulin for pediatric use. I'm also a bit excited about his mention of using Afrezza with artificial pancreas systems. I liked when I read about using the use of AFREZZA with the artificial pancreas systems too. It has been my belief since I read about the "artificial pancreas" that AFREZZA is the most fitting to accomplish the goal at hand, and really, nothing else comes close. So I often wondered why there was never a mention of AFREZZA. Now thankfully, there is! We've got to start gaining momentum some time... |
|
|
|
Post by mnholdem on Jan 3, 2017 15:51:37 GMT -5
Does anybody remember management saying a few years ago that a 2-unit cartridge wouldn't be developed until the pediatric trials? Don't be surprised if they have been working on it.
|
|