|
|
Post by silentknight on Mar 27, 2017 6:57:04 GMT -5
I've said all along that Afrezza has been priced too high. It's one of many problems that MNKD faces in marketing the drug, but basic rules of supply and demand tells you that if a product isn't selling, you drop the price to try and drive sales.
MNKD is shooting themselves in the foot with their pricing strategy. I don't care if the price hasn't been raised in two years. It was too expensive then and it's too expensive now.
Convincing doctors to use it and creating patient awareness is one thing, but pricing is something MNKD has full control over and they're blowing it.
Afrezza already generates next to nothing in sales. Do they think lowering the price would undercut their margins, which are already returning nothing? It's asinine. Apparently, nobody in MNKD has enough common sense to realize any of this. It's astounding.
|
|
|
|
Post by derek2 on Mar 27, 2017 8:51:53 GMT -5
What does this tell us about how MNKD would price EpiHale?
My opinion - and take it for what it's worth (it's free and worth every penny) - is that MNKD has a pattern of mentioning new applications of the Technosphere platform in order to give a good news story and then does little to advance it.
We're 2 years into the "MNKD now has a platform, not a single drug" talk, and no IND, no phase 1 for anything. Talk is cheap.
Also - does anybody know (not guess) what MNKD is trying to get changed on the label? Any link to a transcript talking specifics? Failing that, perhaps we fall back on some of the symposium posters we've seen over the last 2 years. Hypo and speed, through new data analysis (not through new trials).
|
|
|
|
Post by peppy on Mar 27, 2017 9:05:14 GMT -5
What does this tell us about how MNKD would price EpiHale? My opinion - and take it for what it's worth (it's free and worth every penny) - is that MNKD has a pattern of mentioning new applications of the Technosphere platform in order to give a good news story and then does little to advance it. We're 2 years into the "MNKD now has a platform, not a single drug" talk, and no IND, no phase 1 for anything. Talk is cheap. Also - does anybody know (not guess) what MNKD is trying to get changed on the label? Any link to a transcript talking specifics? Failing that, perhaps we fall back on some of the symposium posters we've seen over the last 2 years. Hypo and speed, through new data analysis (not through new trials).
I can give it my best shot. I think MNKD with the 2016 abstracts/clamp studies is going for an ultrarapid classification. my guess.
www.mannkindcorp.com/Collateral/Documents/English-US/Baughman%20poster%20100-LB%20FINAL%20X2.pdf
Use of ultrarapid acting insulin
Type: Grant
Filed: January 25, 2012
Date of Patent: March 21, 2017
Assignee: MannKind Corporation
Read more: mnkd.proboards.com/thread/7479/new-mannkind-patent-published-2017?page=1#ixzz4cXDbDGTQ
The only reason "good" I can think of as to why there are no box configurations on the MNKD website, is, the boxing being redone with ultra rapid designation? In time for the advertisement? ultrarapid? Anyway moving away from that, price for afrezza as shown is the issue.
Epi in every first aide box needs to be replaced. Another fight with a cash cow. RLS, buying MNKD is like the coffee shop buying an expresso machine in the moment. |
|
|
|
Post by seanismorris on Mar 27, 2017 9:44:22 GMT -5
I think there are problems with lowering then raising the price of Afrezza.
MannKind would lower the cost of Afrezza to achieve price parity. But the need to get to parity is because of lack of coverage. (Especially govt)
But, once they do get Afrezza covered then they'd have to raise the price to get back to parity or have margins suffer. The way MannKind (I think) is dealing with this is through coupons and discounts. Unfortunately this means the List price is high, and many docs don't look further.
Personally, I HATE, HATE, HATE coupons, discounts, specials,etc. (I also dislike negotiating) I do all my shopping at Costco and Amazon...
I just want a good/fair/competitive price everyday.
I'd rather MannKind just lower the price (to drive scripts) and worry about the fallout later... but for some unknown reason (idiocy) MannKind doesn't feel the urgency that investors feel.
|
|
|
|
Post by agedhippie on Mar 27, 2017 9:49:50 GMT -5
|
|
|
|
Post by peppy on Mar 27, 2017 9:53:12 GMT -5
aged, how much for a vial of rapid acting mealtime insulin? how how much for a vial of basal? How much are you going through in a month?
|
|
|
|
Post by agedhippie on Mar 27, 2017 10:10:26 GMT -5
Aged, When you buy a vial of rapid acting to fill the pump chamber, what are you seeing on the bill copy sent by the insurer? How much is he insurer saying the retail price is? the discounted price? amt paid?
How long does a vile last you?
Ditto, what is long acting costing a month per invoice?
I don't have a recent invoice for vials but here is my last order of pens which is for three months. I clipped the other half because it has personal information. 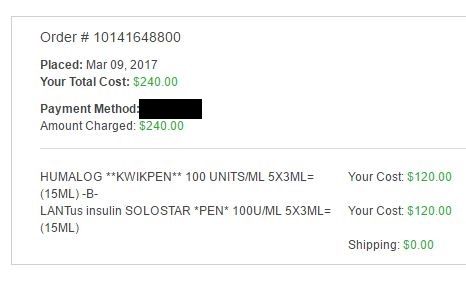 |
|
|
|
Post by agedhippie on Mar 27, 2017 10:16:43 GMT -5
aged, how much for a vial of rapid acting mealtime insulin? how how much for a vial of basal? How much are you going through in a month?
I think our posts crossed  That invoice is for two boxes of Lantus (10 pens) and three boxes of Humalog (5 pens). Over three months I will probably use 8 of the 10 Lantus pens, and all of the Humalog pens. |
|
|
|
Post by rockstarrick on Mar 27, 2017 10:41:12 GMT -5
What does this tell us about how MNKD would price EpiHale? My opinion - and take it for what it's worth (it's free and worth every penny) - is that MNKD has a pattern of mentioning new applications of the Technosphere platform in order to give a good news story and then does little to advance it. We're 2 years into the "MNKD now has a platform, not a single drug" talk, and no IND, no phase 1 for anything. Talk is cheap. Also - does anybody know (not guess) what MNKD is trying to get changed on the label? Any link to a transcript talking specifics? Failing that, perhaps we fall back on some of the symposium posters we've seen over the last 2 years. Hypo and speed, through new data analysis (not through new trials). I resemble that remark !! |
|
|
|
Post by peppy on Mar 27, 2017 10:58:48 GMT -5
Aged, When you buy a vial of rapid acting to fill the pump chamber, what are you seeing on the bill copy sent by the insurer? How much is he insurer saying the retail price is? the discounted price? amt paid?
How long does a vile last you?
Ditto, what is long acting costing a month per invoice?
I don't have a recent invoice for vials but here is my last order of pens which is for three months. I clipped the other half because it has personal information.  Thank you aged. the your cost part... is that what you are paying? Insurance already figure in? Do the quicky for me. What is your rapid acting Humalog costing you a month? $120?
|
|
|
|
Post by zuegirdor on Mar 27, 2017 11:31:32 GMT -5
"[O]ur big prescribers that really loved Afrezza did not come back on Board. And in fact, one of them passed away unfortunately, and that did hurt the Q4 performance." That doesn't make any sense? If they really loved Afrezza why aren't they on board? And why didn't they know right away and correct it? And what patient would drop the drugs they are on because their doctor died? The new rep that handles that area will know who the doctor referred his patients to and make sure the new doc is up to speed. And I'm sure they are working like crazy getting the others back on board. And while I'm at it... I have another question for anyone who wants to answer:-) Are we sure the patients are being told to use a follow up dose? And that people have to buy more Afrezza because of it? ( Fine with me if that's how to get the best results.) Tom's kids don't do that very often. They are not afraid to take enough. And don't need extra. But they are also not trying to be perfect or compete with their numbers. And are the T2's needing more also? I suspect Big Pharma is putting a great deal of pressure on the doctors not to use afrezza. The amount of pressure they put on the FDA not to approve was amazing. Unless they are properly dosing their A1c results won't be better than an RAA so its easy for the doctor to change them back to an RAA.... ....Afrezza is so different from anything current doctors and insulin users have encountered they really have a hard time believing to just "Go Big" with the dose and don't worry. They have allows been taught and learned the hard way the Analogs are dangerous and R insulin is dangerous. Afrezza has to be understood as a new insulin tool. You cannot compare it to RAA PD/PK in terms of dosing. The problem is in the comparison of "units" and timing, pricing, prescription. The whole deal. It is so new and paradigm shattering that its possible that only highly intelligent and motivated patients, with the courage to ignore all the warnings and, frankly, ignorant, dosing claims of physicians and everyone else, have been able to unlock its true potential. Further, every diabetic is different, with different dietary and lifestyle needs. Afrezza use can be customized once you figure out how it works. But many will wait for the paradigm to shift, for the early adopters to blaze the trail and take the "risks" (the risks are less than those you take with RAA!). I see many here are just realizing that the dosing advice per FDA rules is the problem. Yes-follow ups are needed for any meal over 30-40 carbs unless you are getting exercise within a half hour of eating. (Afrezza with a little exercise is AMAZINGLY EFFICIENT!). My kid likes to eat high carb most often 60-100+ g carbs/meal. And he is more sedentary than I would like (computer nerd-the BEST!). Consequently he will even need a third follow up dose! Expensive if you want to eat that way but he is still growing. What we do to save on cost and prevent rises due to "non compliance" (failing to remember to check BG and take follow ups) is use an 8u for first phase after eating (exact time of delay depending upon starting blood glucose level). Simultaneous with that first puff, he takes Humalog shot at 1/2 his normal carb/insulin dose ratio. This usually does the trick. He flat lines the whole meal. Sometimes, three hours later he might still be rising (stomache slower to empty on higher carb meals). A puff of a 4 or 8u easily and quickly stops these before he spends more than half to one hour out of range. What do you all think? Is this regimen too easy? Too hard? Too risky? Good or Bad news for Investors? Should someone be punished for even talking about this? FDA, what do you think about how diabetes is really treated? About people who can improve time in range and simultaneously reduce hypo events? Oh what a mangled clustercluck we have hatched! |
|
|
|
Post by lookingforlogic on Mar 27, 2017 11:37:07 GMT -5
Here is the simplest answer I can quickly come up with using the quote by George Carlin:
Think of how stupid the average person is,
and realize half of them are stupider than that.
The first thing we have to do is totally disregard label warning about spirometry and occasional coughing at the beginning of using afrezza.
Every smoker knows they were coughing after the first cigarette or even second and then never caugh again.
|
|
|
|
Post by peppy on Mar 27, 2017 12:08:12 GMT -5
|
|
|
|
Post by agedhippie on Mar 27, 2017 13:28:57 GMT -5
I don't have a recent invoice for vials but here is my last order of pens which is for three months. I clipped the other half because it has personal information.  Thank you aged. the your cost part... is that what you are paying? Insurance already figure in? Do the quicky for me. What is your rapid acting Humalog costing you a month? $120?
That's what my insurance charges me for my insulin. It's a flat $40 co-pay. My co-pay is capped at $40 per prescription per month through their mail-order pharmacy (you have to use the mail order pharmacy for recurrent prescriptions). |
|
|
|
Post by dreamboatcruise on Mar 27, 2017 13:57:53 GMT -5
What does this tell us about how MNKD would price EpiHale? My opinion - and take it for what it's worth (it's free and worth every penny) - is that MNKD has a pattern of mentioning new applications of the Technosphere platform in order to give a good news story and then does little to advance it. We're 2 years into the "MNKD now has a platform, not a single drug" talk, and no IND, no phase 1 for anything. Talk is cheap.Also - does anybody know (not guess) what MNKD is trying to get changed on the label? Any link to a transcript talking specifics? Failing that, perhaps we fall back on some of the symposium posters we've seen over the last 2 years. Hypo and speed, through new data analysis (not through new trials). More importantly for a company running out of money, talk is not only cheap... it is free. |
|