|
|
Post by matt on Apr 17, 2017 17:04:03 GMT -5
articles.latimes.com/2001/may/31/business/fi-4587Medtronic said it will pay $48 a share for MiniMed, a 9.1% premium over Tuesday's closing stock price, but a near doubling of the $25-per-share price seen in April, before rumors of a MiniMed sale began circulating. There were a couple of deep pocketed companies in the hunt for Minimed at the time, including the company I worked for back then. It was not just about insulin but the full range of drugs that needed to be administered slowly over time including pain medications, antibiotics for HIV patients, and others. Though we had a big pump business, we backed out of the running because it was a difficult electromechanical device to manufacture and that is something where Medtronic excelled and we didn't. I doubt anybody else could have made a business out of it. |
|
|
|
Post by uvula on Apr 17, 2017 21:26:08 GMT -5
articles.latimes.com/2001/may/31/business/fi-4587Medtronic said it will pay $48 a share for MiniMed, a 9.1% premium over Tuesday's closing stock price, but a near doubling of the $25-per-share price seen in April, before rumors of a MiniMed sale began circulating. This is from 2001. I don't understand the relevance to this thread. |
|
|
|
Post by sportsrancho on Apr 17, 2017 22:04:43 GMT -5
|
|
|
|
Post by mango on Apr 17, 2017 23:29:53 GMT -5
dreamboatcruise Can you elaborate on why Mannkind can not do something like this, if they do not make any claim that is not supported by the label? FDA requires that any marketing be balanced and include the adverse effects that occurred in the trials. Small uncontrolled demonstration of use such as what you are suggesting I believe is simply considered incompatible with the requirement to present balanced marketing material... i.e. how do you represent bronchial spasms, hypos, etc. in that sort of setting. I'm sure you've seen many TV ads for other drugs, as it is hard to watch TV without seeing them. The reason they are formatted as they are is because of the requirements of the FDA, not because all the pharma companies hire terrible ad firms. If you pay close attention to the ads you'll see that they don't ever have someone talking about the wonderful results they got. Even when they have an actor play a patient they will have their script say something like "I went to my doctor and my doctor told me xyz MAY help with my... ". It would be great if they could figure out some way of enabling real patient results being shared. Maybe they could organize meetings for people interested in Afrezza and existing Afrezza users. Though if MNKD had any role in organizing such an event, it may still fall afoul of the FDA. • Both of the below TV commercials by Sanofi spend nearly an entire minute talking about risks and side effects. They also employed millions of dollars worth of tactics in order for the viewer to never notice any of them. Only one of these received a warning letter. • Sanofi used these claims in the Toujeo video, Journal: CONSISTENT INSULIN LEVELS with no pronounced peak or wear-off between doses 
PROVEN FULL 24-HOUR BLOOD SUGAR CONTROL

SIGNIFICANT A1C REDUCTION

Sanofi's Toujeo TV Commercial, "Journal"• Sanofi used these claims in the Toujeo video, Mr. Groove: BLOOD SUGAR-LOWERING ACTIVITY FOR 24 HOURS AND BEYOND 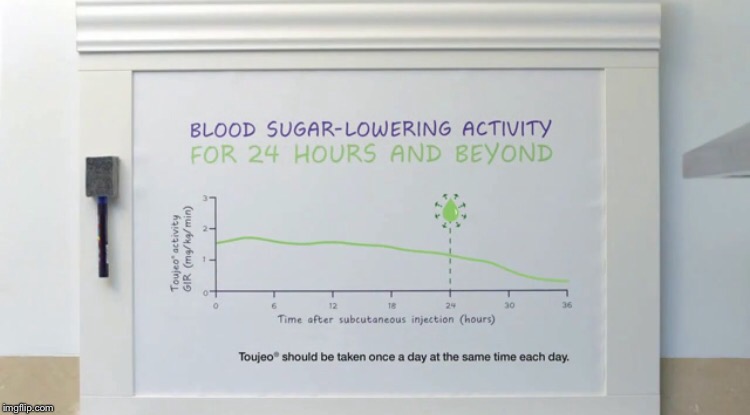
PROVEN BLOOD SUGAR CONTROL ALL DAY AND ALL NIGHT

SIGNIFICANT A1C REDUCTION

Sanofi's Toujeo TV Commercial, "Mr. Groove".• What did the FDA have to say about, Mr. Groove? Not much. The FDA issued a measly "warning letter" to Sanofi, which is just one of many Sanofi has received from the FDA throughout the years. This is the least serious of them all, in fact. The FDA Warning Letter to Sanofi—Toujeo TV Commercial
FDA—Sanofi's Promotional Materials Recommendations for the Toujeo TV Commercial• For those wondering, here is a 39 minute and 41 second video explaining how to inject Toujeo:
How to Inject Toujeo |
|