|
|
Post by mnkdfann on Jan 15, 2021 16:28:21 GMT -5
I am struck by the common thread of Novartis/Sandoz running through MNKD's team and recent activity, and the fact that they seemed to have emphasized/strengthened their pediatric expertise. They looked to be well prepared for the pediatric trial and one has to wonder if Novartis might not be involved. Qrum: Thomas Hofmann - Pediatric Pulmonologist - Prior experience included Chief Scientific and Safety Officer for what was to become Novartis Germany - also member of the team that gained approval for inhaled tobramycin Novartis owns Lamprene (clofazimine) Co-promote with Vertice Pharma: - Scott Meyers (CEO) - most recently as Vice President, General Manager, Dermatology of Fougera Inc., a Sandoz / Novartis company - Prior to Fougera, Scott led Strategic Planning & Market Analytics for Sandoz US- Noel Grennberger (CCO) - most recently Executive Director of Respiratory at Sandoz U.S., the generics division of Novartis, and was responsible for building and commercializing Sandoz’ portfolio of respiratory medicines MNKD: James Shannon - prior global head of Pharma development for Novartis - recent assumption of chairman role M Castagna - prior to Amgen, former VP and head of Biopharmaceuticals NA for Sandoz (generic arm of Novartis)Is that game starting up again (big pharma connections to Mannkind, hinting at great things underway)? Is Novartis the new Amgen, this time around?  |
|
|
|
Post by mnkdfann on Jan 4, 2021 8:57:51 GMT -5
Anyway, if you go to the Napa area ... make sure you look for a tiny sign next to a road that goes up into the forest. There you will see the castle ... called the Castello di Amorosa. Does the wine have a smoky after-taste? youtu.be/VafRHe1rIQo |
|
|
|
Post by mnkdfann on Dec 30, 2020 10:52:58 GMT -5
Adding Qrum to the agreement with MidCap as a borrower. I'm not a lawyer, but it reads as though Qrum was already listed in a prior revision of the Existing Credit Agreement. This latest looks to be about correcting a name in it (correcting a legal typo)? "Amendment to Existing Credit Agreement. Subject to the terms and conditions of this Agreement, including, without limitation, the conditions to effectiveness set forth in Section 8 below, the Existing Credit Agreement is hereby amended as follows: (a) The definition of “Qrum” in Section 15 of the Existing Credit Agreement is hereby amended and restated as follows: ““Qrum” means QrumPharma, Inc., a Delaware corporation.” Anyway, it doesn't look to me as though this is going to move the SP in any big way today.  |
|
|
|
Post by mnkdfann on Dec 29, 2020 19:24:20 GMT -5
So it looks as though you use the trade date for tax purposes, whereas we (here in Canada) use the settlement date. Now, how can I take advantage of that?  |
|
|
|
Post by mnkdfann on Dec 29, 2020 19:00:41 GMT -5
Not selling anything...well I sold NIO. That’s all I can afford to pay taxes on. Have carryover losses anyway. Those profits from NIO wil go into Mannkind next year. I think we may see a correction the end of January so probably add in March for my April-November run:-) Already have 2022 calls that are ITM. In Canada, at least, taking into account trade settlement days, next year's trading actually starts tomorrow. is it different in the States? |
|
|
|
Post by mnkdfann on Dec 29, 2020 16:46:11 GMT -5
Down a years income Today. Bought as much as I could as well and am smiling ear to ear. Next year will be amazing. Yet you look blue. (FWIW, I'm colour blind. I think his avatar is blue. If not, it still looks blue to me.) |
|
|
|
Post by mnkdfann on Dec 28, 2020 16:00:29 GMT -5
I'm just curious what other Pharma UTHR purchased this voucher from. Anyone here know? I didn't see it named in the filing, but I may well have missed it.
|
|
|
|
Post by mnkdfann on Dec 28, 2020 10:00:40 GMT -5
Not really......Selling PRVs are pretty common. Think about it...if a company is granted a PRV that means they have a good drug in trials but there are times when the drug fails and the company has a useless voucher so they see if there's a market for it. Willing SELLER Willing BUYER. That makes a market. Seems Legit. Legit but the process seems sleazy. If a company has a drug for a Rare Pediatric Disease they can get a "Rare Pediatric Disease Priority Review Voucher" directly from the FDA (presumably without a huge payment). This sounds fine. If UTHR has a drug for a Rare Pediatric Disease, why can't they get a voucher directly from the FDA? If the voucher is rare (i.e. limited supply), how are they given out? Is it random, based on need, or based on cozy big pharm relationships? If the original company can't use the voucher, shouldn't it be returned to the FDA so the FDA can give it to someone else? This is almost like someone on a kidney transplant list selling their spot to someone else. This is good for UTHR or they would not have done the deal. But it would be interesting to know more about how the process works. |
|
|
|
Post by mnkdfann on Dec 26, 2020 19:34:13 GMT -5
Hello everybody and Happy Holidays! I have been extremely busy for the past six months as my group of animal healthcare companies have been exploding! USA, UK, Australia, New Zealand has been phenomenal and we’re now expanding into Asian and South America markets. I think I’m putting in as many hours these days as the legendary Alfred E. Mann used to. It’s not uncommon for me to work 60 hours per week, which doesn’t leave me any spare time to peruse any stock discussion boards. Is that a publicly listed animal healthcare company, or private? |
|
|
|
Post by mnkdfann on Dec 17, 2020 19:03:30 GMT -5
I'm surprised by this too!  I don't see the value in this arrangement between the two companies. At least they're not hawking some other Pharma's brand of insulin. |
|
|
|
Post by mnkdfann on Dec 17, 2020 18:37:11 GMT -5
Simply outstanding. This has been shaping up to be a December to remember. Isn’t that a car commercial ad? :-) The one on SNL last weekend? I just watched that like an hour ago. It was very funny.  |
|
|
|
Post by mnkdfann on Dec 17, 2020 17:50:49 GMT -5
Unless I dreamt it, Mannkind (e.g. Castagna) has talked about doing such co-promotions (at Annual Meetings and the like) through its sales force off and on for over 2 years.
So glad they finally landed a fish.
|
|
|
|
Post by mnkdfann on Dec 10, 2020 12:52:14 GMT -5
"About MannKind Corporation MannKind Corporation (Nasdaq: MNKD) focuses on the development and commercialization of inhaled therapeutic products for patients with endocrine and orphan lung diseases. MannKind is currently commercializing Afrezza® (insulin human) Inhalation Powder, the Company’s first FDA-approved product and the only inhaled ultra rapid-acting mealtime insulin in the United States, where it is available by prescription from pharmacies nationwide." What does "commercializing" mean? It must mean something different than "selling" or "manufacturing". They may be focussing on the 'make a profit' part of the definition.  com·mer·cial·ize /kəˈmərSHəˌlīz/ Learn to pronounce verb gerund or present participle: commercializing manage or exploit (an organization, activity, etc.) in a way designed to make a profit. |
|
|
|
Post by mnkdfann on Dec 10, 2020 12:30:24 GMT -5
This graph is from the first link: 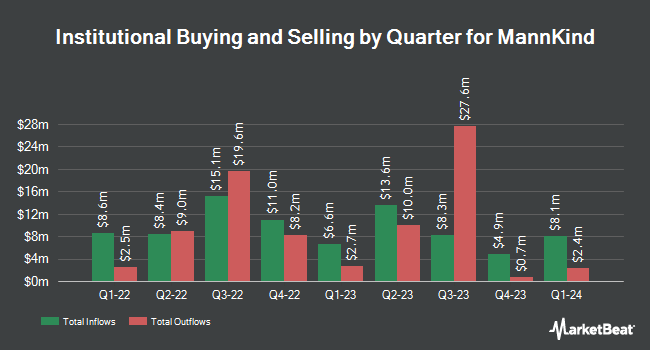 |
|
|
|
Post by mnkdfann on Dec 7, 2020 20:40:37 GMT -5
|
|