|
|
Post by agedhippie on Jun 26, 2023 9:07:24 GMT -5
Or a broken trade.
|
|
|
|
Post by agedhippie on Jun 26, 2023 9:05:35 GMT -5
I've been wondering how launch of Inhale-3 can be consistent with the goal of CFBE for the endocrine business unit (EBU) this year. Inhale-3 has to be expensive. It appears that expenses for clinical trials go under R&D. Then, are R&D expenses for this trial not counted against cash flow or profitability of the EBU? I would have thought that this would need board sign-off in which case it may have it's own budget line under R&D (although I have seen this sort of thing assigned to sales and marketing before  ) This is what I would expect, but I have always been the beneficiary of the budget rather the the person who has to raise it... |
|
|
|
Post by agedhippie on Jun 26, 2023 9:02:22 GMT -5
This can be powered to show clinical as well as statistical significance with 120 patients. At some point, the cost of adding more subjects has to be weighed against the benefit. Clinical trials are hugely expensive. The goal is to size the study properly. It's a Goldilocks problem. More is not always better unless you expect large numbers of study dropouts. To be honest, I doubt that MC had much to do with designing the study other than looking at the cost numbers. Excellent points. I especially like the acknowledgement that senior executive leaders run a business and that technical details are often the domain of folks who work for the executives. It’s one of the reasons I’ve laughed at the brouhaha over documents being places they oughtn’t as though the celebrity in the spotlight had carefully reviewed and selected tons of paperwork to take home. More likely folks tasked with assembling the material asked the celebrity where they’d like the dozens of boxes stacked and the decision-making ended there. Whether it’s lawyers, paralegals, interns, assistants, researchers, or engineers, the executives have people who do stuff for them. They have to. This is completely true. The Mannkind involvement will be at a very high level, primarily goal setting. Ensuring the trial is properly powered to resolve those goals is the job of Jaeb. Companies like Jaeb exist to design trials and manage logistics, and Jaeb is one of the better ones. |
|
|
|
Post by agedhippie on Jun 26, 2023 7:04:52 GMT -5
For T1s I am always assuming they are using a CGM. If they are not, well, they are probably not too worried about great control nor hypos. And just to be clear on this. Good control is the control that you want, it's not what someone choses for you. That's fundamental - it's your life, not theirs. |
|
|
|
Post by agedhippie on Jun 26, 2023 6:56:49 GMT -5
If you want control post 2 hours you don't want a prandial. The non-diabetic within two hours should be back to baseline. For all diabetics the goal should be sub-140 in 2 hours. If you want to argue the RAA is better than afrezza after 2 hours what I would say is your RAA sucks. It should be out of your system in 2 hours. This is the advantage of the AID. Its a post 2 hour tool. The whole point of the study is nothing beats afrezza for meal time control. Its also provide a great alternative for T1s who want to ditch the pump but who still want great control. I'm not the one saying RAA is better after the two hour mark Mannkind is saying it, that's was a link to a chart on their web site. Afrezza is not getting you back to 140 in two hours, it's leaving you at about 170. It would be interesting to see what the chart looks like for the following two hours but understandably they don't seem to be in a hurry to show that. At the end of the day the primary outcome measurement they chose was change in A1c, and the best result came from using an AID pump and not adding Afrezza. Notice that they haven't published TIR although they have that data. |
|
|
|
Post by agedhippie on Jun 25, 2023 15:31:45 GMT -5
If we want less control but no daily injects the answer is icodec and afrezza. How about icodec Afrezza plus a CGM FOR BETTER CONTROL… I would always advocate for a CGM because you need to make informed decisions, even if the decision is that you are not going to ignore a high for the moment. It's letting you take control of your diabetes. Mannkind used CGMs in the ABC trial, but they haven't published those results. I don't really see why Icodec would give significantly better control than Tresiba. The problem always used to be that Lantus wore off in less than a day so you ended up having to split dose, Toujeo and Tresiba both last over a day so that is fixed. The reason the market thinks Icodec could be a big deal is because T2 diabetics on basal could inject weekly rather than daily which would reduce skipped doses (T1 diabetics don't have the option to skip, but the option to dose weekly is welcome!) |
|
|
|
Post by agedhippie on Jun 25, 2023 9:34:22 GMT -5
The take away is nothing is better than afrezza for meal time control. What this shows is afrezza beats the AID for this. Because of its speed of action there is a reduce chance of hypos which allows a reduction in baseline BG. The question is how aggressive can you get with a T1 and of course that will depend on the person. Buts lets say we can get so aggressive we can target 90 with MDI. the obvious question is what is the AID bringing to the party then? If we want the best A1c the answer is the AID with afrezza. If we want pretty great control and no pump hassle the answer is MDI with afrezza. If we want less control but no daily injects the answer is icodec and afrezza. I know there is a huge pump business but most people really don't want to wear them, especially the kids - if they don't need to. With afrezza, they don't need to. This study is saying nothing different than what we have been saying right here on proboards for years. Mike just needs to get the large scale study done. Look at the chart again. If you are looking at the first two hours Afrezza is greatly superior, if you are looking at three hours and further then RAA is superior. Of course you can compensate by taking a second Afrezza dose but now you are taking insulin twice as often as you would with RAA. Maybe you can be aggressive with Afrezza and target 90 with MDI, maybe you can do the same with an AID pump. Until the outcomes are proven in a trial it's all maybes on both sides. Lol. The chart in the paper from The Afrezza ABC trial quite clearly shows that if you want the lowest A1c then you should use an AID pump alone. The gradient of reduction for the AID pumps is steeper than either of the Afrezza options, either AID for basal, or Tresiba. So people don't have to go and dig through the ADA presentations here is the chart:  What matters here is the gradient since that is the amount of reduction in absolute terms from start to finish, the steeper the gradient the better. Two things stand out from this; Using Afrezza will reduce your A1c (no surprise there for us), and that it underperforms the control arm which is just the pump. Actually this is not a trial I would think MNKD should do since the study is not good. The whole point of studies is to pick good trial subjects and avoid bad ones. |
|
|
|
Post by agedhippie on Jun 24, 2023 13:00:48 GMT -5
What I find interesting is all 3 groups showed an almost equal drop in HbA1c at 90 days was this due to controlled meals like in part1 ? It's not unusual in a trial. People tend to be more careful, and also there is a lot more follow up (nagging) than you get from the normal medical staff because they want to make sure you are following the treatment as described. This is one of the reasons why having a reference arm is so important because you want to be sure the improvement is due to the treatment and not just the management protocol. It's also why real world numbers where there are not an army of trial managers standing over people are important. |
|
|
|
Post by agedhippie on Jun 24, 2023 8:04:14 GMT -5
OK butt the issue is not A1c nor is it TIR as currently defined. Its post prandial control. Its about getting back under 140 in 2 hours in the best case. This is why I think Irl Hirsch will be focusing on 2 hour PPG in the study and working to redefine things. The thing is only afrezza can do this so he has his work cut out for him. As we all know A1C is an average like average miles per hour on a trip. The 2 hour PPG is like going through a 45mph area at 65 and getting a ticket. Let's have a look at the trial filing: Primary Outcome Measures :
Change in HbA1c [ Time Frame: 90 days ]
Change in glycated hemoglobin (HbA1c) from baseline to end of study
So actually the issue is A1c.  Interestingly one of the secondary outcome measures is TIR but they don't seem to be talking about that. |
|
|
|
Post by agedhippie on Jun 24, 2023 7:52:59 GMT -5
Running trials is what Jaeb does so I am pretty certain that they will design the trial properly. They have done huge trials in the past so they have all the contacts.
|
|
|
|
Post by agedhippie on Jun 24, 2023 7:26:26 GMT -5
Why are they not good? This seems pretty great and the reason for the large study for 2 hour ppg control. ... Did you look at the part 2 graph? It's pretty obvious what the problem is. The HbA1c drop for TS + Tresiba and TS + AID is the same so they are basically equivalent. The drop for the AID pump alone is larger meaning that day to day the AID pump used alone outperforms the other two. I would have expected TS + AID to outperform TS + Tresiba if the idea that the Afrezza weakness in earlier trials was due primarily to the basal insulin, but that didn't happen. That's before we consider the differing starting points. There is a reason why Mannkind always chose the 2 hour PPG. It's because at 3 hours there is no difference, and beyond that RAA is better. This is why things like the HbA1c results are taken more seriously than 2 hr PPG as there is decades of data directly relating HbA1c to complications. This is the chart from the Mannkind website: 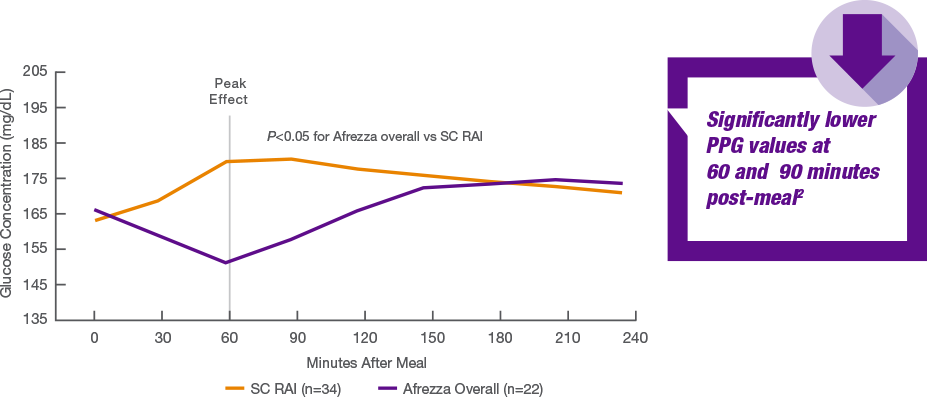 |
|
|
|
Post by agedhippie on Jun 24, 2023 5:04:35 GMT -5
|
|
|
|
Post by agedhippie on Jun 23, 2023 13:39:58 GMT -5
Can’t believe MNKD has not released any news. WTF is Castagna doing??? You have LQDA surfing because they have the big boys of Wall Street backing them. WTF LQDA is a speculative stock, and right now the back of the envelope calculation looks reasonable - they are going to split the DPI market with UTHR and especially the PH-ILD market. |
|
|
|
Post by agedhippie on Jun 23, 2023 13:24:20 GMT -5
Year to date the S&P Biotech ETF is up 3%, UTHR is down 19%, and MNKD is down 25%. Largely MNKD seems to be tracking UTHR with occasional breakouts, but reverting to tracking.
|
|
|
|
Post by agedhippie on Jun 22, 2023 16:55:54 GMT -5
Another useless small study to show share holders Mike's doing his job and justifying his pay and reinforcing job security. Not in this case. |
|