|
|
Post by sportsrancho on Feb 9, 2017 10:15:27 GMT -5
I like the idea of delivering directly to the patient. Yes, added shipping cost, but cut out distributor and pharmacy cost in the final product. Should make it more attractive to insurers. Also simpler for the patient, and it assures the med gets refilled if on auto-delivery. I love it! |
|
|
|
Post by peppy on Feb 9, 2017 10:33:08 GMT -5
cutting out the distributor.... does that mean cutting out the pharmacy/pharmacies?
What is the consequence of that?
|
|
|
|
Post by liane on Feb 9, 2017 10:39:57 GMT -5
My take is that the distributors and pharmacies are still in the picture for those patients that prefer that mode of delivery. But for patients that accept direct delivery, the cost to the insurer can be reduced as these 2 entities do not have to get a cut. So I would think the insurers could conceivably put Afrezza on a lower tier in exchange for direct delivery.
|
|
|
|
Post by mnholdem on Feb 9, 2017 12:44:08 GMT -5
The direct mail pharmacy is more reason for MannKind to do a study to prove that Afrezza doesn't need to be refrigerated.
|
|
|
|
Post by johntherancher on Feb 9, 2017 13:50:24 GMT -5
According to the FDA, "Does the FDA review and approve all advertisements for drugs before their release? No. In most cases, federal law does not allow the FDA to require that drug companies submit ads for approval before the ads are used. We see many ads at about the same time the public sees them. Many drug companies voluntarily seek advice from us before they release TV ads. However, if we believe that an ad violates the law, we send a letter to the drug company asking that the ads be stopped right away." Access FDA website for more information: www.fda.gov/Drugs/ResourcesForYou/Consumers/PrescriptionDrugAdvertising/ucm076768.htm#approve_advertisementsGiven the, IMO, dire circumstances MNKD is now in based on limited resources and time, we can't afford to wait for the nonexistent "FDA approval of the commercial". It will take some time, but it should not take 3 to 4 months to get the commercials out on the street. My advice to Matt is to do it already. Thanks, I feel better already. |
|
|
|
Post by careful2invest on Feb 9, 2017 14:24:55 GMT -5
I like the idea of delivering directly to the patient. Yes, added shipping cost, but cut out distributor and pharmacy cost in the final product. Should make it more attractive to insurers. Also simpler for the patient, and it assures the med gets refilled if on auto-delivery. That would be a cost effective and efficient method to possibly adopt. But in addition, Advertising just needs to happen! Very few people, pwd, doctors have even heard of this revolutionary drug, AFREZZA! As much money as Alfred Mann put in the development and approval, why doesn't the Alfred Mann Foundation do what it takes to get AFREZZA across the finish line? They are in this deep! Let's get it done! Instead of dilution and a reverse split, beating the hell out of the pps and the retail investor... Again!!GLTA!! |
|
|
|
Post by sportsrancho on Feb 9, 2017 14:37:43 GMT -5
The direct mail pharmacy is more reason for MannKind to do a study to prove that Afrezza doesn't need to be refrigerated. Yes! And let's get that Afrezza App!! |
|
|
|
Post by mnholdem on Feb 9, 2017 15:11:44 GMT -5
While I would really like to see tons of Phase IV clinical trials, I am aware that registering many clinical trails with the FDA would cost MannKind a small fortune. For example, MannKind's recent application for review of clinical trial data for approval of an Ultra-Rapid Acting classification on the label would likely fall under the review submission type Supplement with Clinical Data, the PDUFA fee for which would have been $395,700 in 2015 according to the table below. High costs of dealing with the FDA are why MannKind is being selective in registering trials to gather relevant data, especially in light of their financial situation. Also, PDUFA fees are only one part of the cost of clinical trials. There are also patient reimbursement costs (i.e. travel expenses), costs associated presenting the data from clinical trials and other costs.
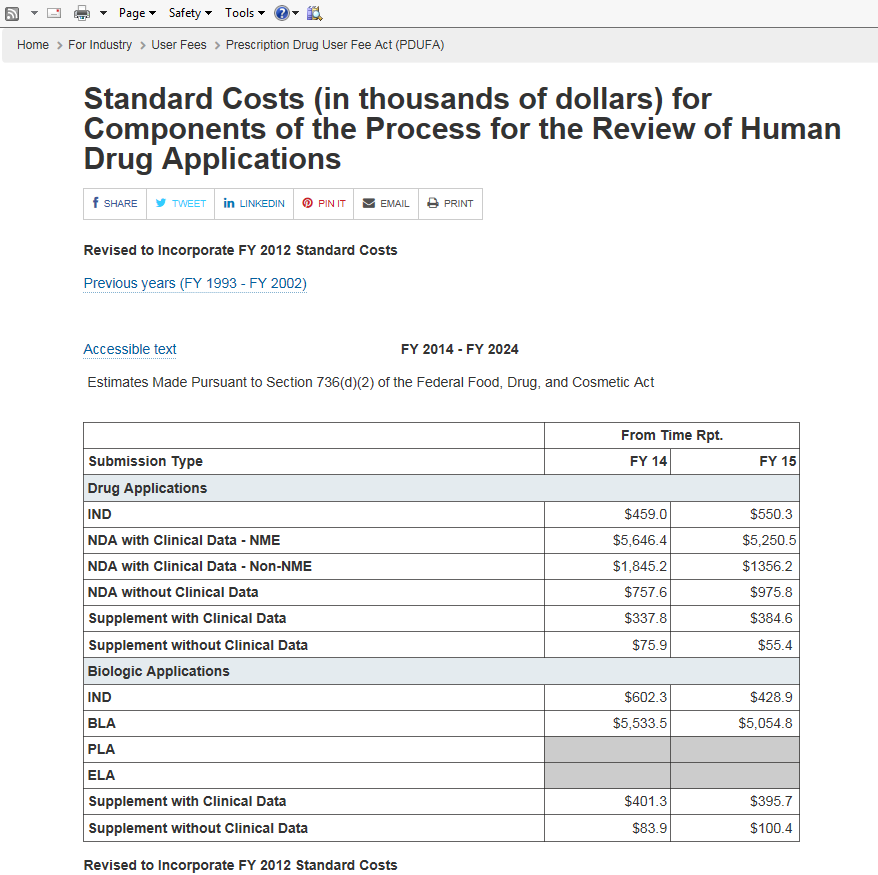
It's certainly expensive to prove a drug is effective these days. 
Source: www.fda.gov/ForIndustry/UserFees/PrescriptionDrugUserFee/ucm093484.htm
|
|
|
|
Post by peppy on Feb 9, 2017 15:18:53 GMT -5
Pardon my French, consider the present expense for FDA services is part of the gig.
Only big pharma will have the money to get drugs approved.
|
|
|
|
Post by markado on Feb 9, 2017 21:36:46 GMT -5
How about some slogans or pitch points for a national ad-
Afrezza - demand more and less from your diabetes care.
More time in range
More rapid onset and quick response
More privacy
More convenience
More freedom
Less needles and injections
Less likelihood of hypoglycemic events
Less likelihood of site infection
Etc. Etc.
Talk to Diabetics. Learn what they'd like to hear and cross that with what you can say without inviting over-scrutiny.
Again, Mike C, the content and concepts above are free for your use, at your discretion.
|
|