|
|
Post by hidanm on Jun 1, 2017 5:28:33 GMT -5
Hi guys, In your opinion, what will be the target price after these agreement?
thanks
|
|
|
|
Post by hillsave on Jun 1, 2017 5:34:26 GMT -5
We've only just begun to breathe
|
|
|
|
Post by therealisaching on Jun 1, 2017 6:25:31 GMT -5
Who else is calling after Brazil? UAE? China? Margin?  |
|
|
|
Post by tw12 on Jun 1, 2017 6:35:49 GMT -5
My quick guess: Europe, Japan, Israel, in that order.
Perhaps someone (much more techno-fluent than I) set up a survey graph?
|
|
|
|
Post by mnholdem on Jun 1, 2017 6:44:05 GMT -5
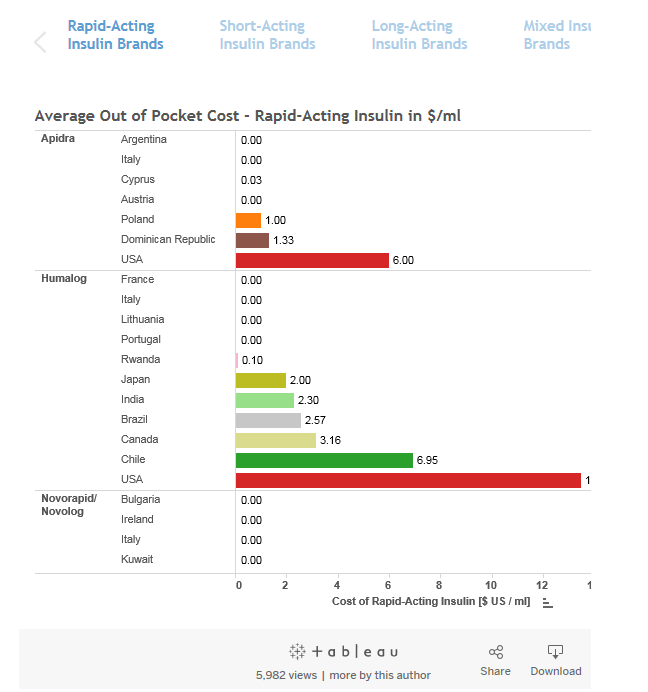
The out of pocket cost for rapid-acting insulin in Brazil is approximately 1/4 of the OPC in the USA.
|
|
|
|
Post by matt on Jun 1, 2017 6:58:41 GMT -5
The one thing that someone else already mentioned is that the distributor will be responsible for pricing. I hope they price it competitively, unlike SNY. Pricing is subject to government approval, so expect something of a roller coaster ride on pricing. Brazil has a history of high inflation and while the dark days of several thousand percent hyperinflation are over, the rate is still high by US standards, and periodic imposition of currency controls sometimes means that payments for imports can be slow which erodes the value of any sales. An experienced partner knows how the game is played and will constantly tweak prices to keep up, but it is a bumpy road. In some years you can make a small fortune in Brazil, and the next nothing at all. Overall, it is a worthwhile place to do business but it has always been unpredictable. The requirement for more stability studies is 100% believable; it is incredibly hot and humid, but the trade-off is incredibly skimpy bikinis.  You take the good with the bad. |
|
Deleted
Deleted Member
Posts: 0
|
Post by Deleted on Jun 1, 2017 7:00:11 GMT -5
What is especially exciting about this deal is that, unlike MannKind here in the USA, Biomm is known and has been respected by the Brazilian medical community for 40+ years. Because Biomm is so well known and were the #1 insulin manufacturer & distributor in Brazil (#4 globally) once they launch Afrezza, I believe you will see very rapid sales growth. They won't face the obstacles MannKind has had to overcome. Great choice, MannKind! According to Osborne, this was "a deal for the sake of a deal because the companies needed to show something". Lol |
|
|
|
Post by hillsave on Jun 1, 2017 7:11:28 GMT -5
My quick guess: Europe, Japan, Israel, in that order. Perhaps someone (much more techno-fluent than I) set up a survey graph? Japan will be last. Very long process. CANADA would come before Japan |
|
|
|
Post by mnkdfann on Jun 1, 2017 7:27:43 GMT -5
The out of pocket cost for rapid-acting insulin in Brazil is approximately 1/4 of the OPC in the USA. The problem with the comparison is that the average monthly wage in Brazil is even less than 1/4 of what it is in the USA. |
|
|
|
Post by peppy on Jun 1, 2017 7:28:08 GMT -5
The one thing that someone else already mentioned is that the distributor will be responsible for pricing. I hope they price it competitively, unlike SNY. Pricing is subject to government approval, so expect something of a roller coaster ride on pricing. Brazil has a history of high inflation and while the dark days of several thousand percent hyperinflation are over, the rate is still high by US standards, and periodic imposition of currency controls sometimes means that payments for imports can be slow which erodes the value of any sales. An experienced partner knows how the game is played and will constantly tweak prices to keep up, but it is a bumpy road. In some years you can make a small fortune in Brazil, and the next nothing at all. Overall, it is a worthwhile place to do business but it has always been unpredictable. The requirement for more stability studies is 100% believable; it is incredibly hot and humid, but the trade-off is incredibly skimpy bikinis.  You take the good with the bad. I know stability studies need to be done. I do see the humor, that one stability study has been done, and like most things with afrezza, it was done by a user.
During the early stages of Afrezza’s development, it was claimed that it could go without refrigeration for more than two months. However, the packaging instead says to only store at room temperature for 10 days. This extreme test goes 16x longer than the labelling to see how it still performs.
Since starting on Afrezza I have been wondering if Afrezza really does require refrigeration. As you will see in the video, where I stored the insulin was certainly much warmer than average room temperature.
The blister pack spent 161 days without refrigeration before this test. It was first put in the heated hothouse on August 22nd, and then taken out on December 5th (105 days). After that, it spent 56 days in the hottest room in my house during a typically hot Australian summer. Melbourne had 20 days above 90° F (32.2° C), and 5 days above 100° F (37.8° C) during the test period.
Unlike the US, summer officially starts in Australia on December 1st. For an official temperature record of Melbourne over this period, visit this Bureau of Meterology page.
To test if Afrezza still worked, I used a can of Australian full strength Coke, (which has sugar as 100% sucrose in Australia). Sucrose and caffeine together make this drink raise glucose levels quickly. A control test using newly-ordered Afrezza stored in the fridge was also conducted (video will be uploaded soon). Basal insulin was the amazing Tresiba, and no food or other insulin had been taken prior to the tests.
Long story short, there was NO decrease in Afrezza’s efficacy after the five months. It worked flawlessly and glucose never left the non-diabetic levels. Glucose started at 4.2 mmol/L (75 mg/dL) and ranged from 3.8 mmol/L (68 mg/dL) to 4.6 mmol/L (83 mg/dL) during the test. I certainly won’t be requesting refrigerated shipping from the US anymore.
The significance of this really can’t be overstated. The Frio insulin cases we use when travelling barely work in tropical humidity. For diabetics who travel frequently, those who live in hot climates, or anyone who doesn’t want a fridge packed full of insulin, this is a game-changer.
Put simply, Afrezza does not need refrigeration and there is no other insulin on the market that can make this claim.
The manufacturer, Mannkind should capitalise on this feature, and submit revised storage conditions for approval. Extended room temperature storage is yet another major point of difference between Afrezza and all other insulins.
Matt
afrezzadownunder.com/2016/01/afrezza-extreme-storage-challenge/
Brazil, girls in bikini's and men that can reach their feet.
|
|
|
|
Post by agedhippie on Jun 1, 2017 8:18:57 GMT -5
The out of pocket cost for rapid-acting insulin in Brazil is approximately 1/4 of the OPC in the USA. A quick back of the envelope calculation puts that Brazil price at roughly the same as the UK price so I think it's near enough. |
|
|
|
Post by hellodolly on Jun 1, 2017 8:31:46 GMT -5
Where does it say that Afrezza has not been approved or is required to go through this? |
|
|
|
Post by hellodolly on Jun 1, 2017 8:34:39 GMT -5
9 months? Is that with or without fast-track? www.jmcinc.net/brazil-oks-fast-track-assessment-for-device-and-pharma-trial-applications/Brazil OKs fast-track assessment for device and pharma trial applications by JMC on July 6, 2012 Brazil’s regulatory agency, Anvisa, has announced that proposals to fast-track the licensing-assessment of a large proportion of applications for clinical trials of drugs and medical devices have received approved1. Pending the formal publication of the enacted resolution by Anvisa and the precise detail of the policy, the government has issued a statement confirming that applications involving trials already approved in the EU, US, Japan, Australia or Canada will be fast-tracked, said Anvisa… The partner may already have it approved since diabetes is it epidemic proportions in Brazil. BIOMM is a known insulin producer in the country. It's their business. I bet you they get this fast tracked if not handled already. Exactly, the source doesn't say Afrezza in particular has not been approved. It's just a source that gives a very brief overview of the approval process on drugs in Brazil...soft bashing by the poster. |
|
|
|
Post by ptass on Jun 1, 2017 8:35:45 GMT -5
The out of pocket cost for rapid-acting insulin in Brazil is approximately 1/4 of the OPC in the USA. The problem with the comparison is that the average monthly wage in Brazil is even less than 1/4 of what it is in the USA. Maybe they intend to use the insulin they in cold storage to lower their production cost. I know they have a contract with Amphastar but does anyone know if this is limited to US sales. |
|
|
|
Post by mnkdfann on Jun 1, 2017 9:02:20 GMT -5
The partner may already have it approved since diabetes is it epidemic proportions in Brazil. BIOMM is a known insulin producer in the country. It's their business. I bet you they get this fast tracked if not handled already. Biomm develops and licenses technologies. It does not (as far as I can see from its web page etc.) commercially produce insulin itself. I believe the producer was the company Biomm SA split from, i.e. Biobras. Novo purchased Biobras and its insulin production facilities circa 2002. Nonethless, Biomm certainly has expertise getting things approved. But I doubt they would have done the approvals in advance of a deal. In fact, by what right / by what authority would they have to get approval for someone else's product prior to an agreement being in place? And if there was such an agreement, surely it would have had to have been announced previously. The agreement announcement said: "MannKind will manufacture and supply Afrezza to Biomm, and Biomm will be responsible for promoting and distributing Afrezza within Brazil" So (according to that) Biomm is not doing the producing. |
|