|
|
Post by agedhippie on Feb 24, 2023 15:29:19 GMT -5
Here is what he said on One Drop - "And long term, I just think -- I look at what One Drop has done with data, collecting data sets and predicting outcomes over time. I think this is where we want to go." IDK about "where we want to go", because afrezza is different. There is no carb counting. ... You could be right that there wouldn't be anything useful to learn, however I tend to think we could learn more about timing and splitting of bolus from much larger data sets with CGMs outside of a trial setting.... If there is an idea that you can take Afrezza without carb counting then there should be a trial run to prove it because there is not a bat in hell's chance of the FDA approving that as it stands. It's worth noting that the last time they ran that trial all the Type 1s crashed and burnt - they had to withdraw the Type 1 diabetics from the trial on safety grounds. Why does this real world data matter? Look at the Medtronics paper on that same dataset for their 780G pump. They could prove for the 4,000 people who uploaded data there was a TIR of 76%. That's a solid evidence and endos like it because it shows them what to expect. If Mannkind could do the same thing it would go a long way to proving the value of Afrezza. That's why Mike likes the idea. Beyond that as ktim says it allows you to see infer suitable dosing patterns from the data. Anything that clarifies this with evidence can only help promote Afrezza. |
|
|
|
Post by agedhippie on Feb 23, 2023 22:57:15 GMT -5
If you want to see what the Dexcom integration would probably look like see the InPen app which uses that Dexcom API (InPen is a Bluetooth linked insulin pen so almost exactly like bluhale)
|
|
|
|
Post by agedhippie on Feb 22, 2023 18:09:29 GMT -5
I told Mango earlier you would not like this. I wouldn't say wet blanket I would say your argument is more like a wet noodle. The entire purpose of the AID is to provide better control in the 2 hour period after meals and to attempt to stop the spike to avoid the out of control highs. Tresiba is not doing that. Afrezza is. ... Did you read what I wrote because you don't seem to understand how AID works. The AID pump did exactly what is should have done which not to change any settings because you had bolused so it had sufficient insulin to cover the carbs. Hence you are replicating that STAT graph. Afrezza starts faster than RAA. That's not exactly news and why no endo will care. As to the AID performance, they understand the concept of insulin on-board. |
|
|
|
Post by agedhippie on Feb 22, 2023 16:36:16 GMT -5
Here comes your favorite wet blanket  I have no idea why anyone thinks this is a surprise unless they don't understand how AID pumps work. TL;DR You have bolused for the meal and hence have insulin onboard so the pump (correctly) will not adjust the basal rate. Therefore the result should be the same as for STAT - you are looking at RAA bolus vs. Afrezza and already STAT told us what that outcome will be. Longer version You are looking at a maximum of 120 minutes. We already know from the STAT results that what the 120 minute readings will be: 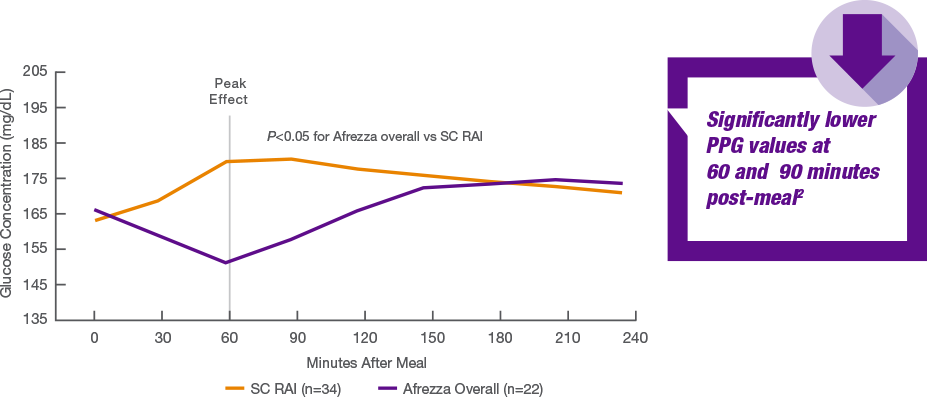 Why does anyone think this is going to different for an AID pump over Tresiba? The AID pump provides basal via RAA, and because the pump knows you have bolused it know there is insulin onboard to cover the food so it's not going to change the basal RAA and you get a flat basal curve, just like you would with Tresiba. This is a definitely not going to move the endos. The large scale ABC trial will be interesting, especially the Afrezza with food and pump the rest of the time arm. |
|
|
|
Post by agedhippie on Feb 20, 2023 11:02:35 GMT -5
[Tweet about ShareIntel being hired by NVVE, VERS, VRSSF, and HOTH] I am fascinated to see what the outcomes from these investigations will be (apart from a stack of money for ShareIntel.) When do we expect them to name names and file lawsuits? |
|
|
|
Post by agedhippie on Feb 20, 2023 10:28:56 GMT -5
Aged - again you are right - almost. V-Go is suppose to get coded as HCPCS code A9274 for External ambulatory insulin delivery system. This falls under Plan D. The thing is I am not sure it always is and it seems sometimes they are coding it as an insulin pump, IDK. To be honest I don't really care much about V-Go especially since Mike just admitted it will never be a big seller. ... The funny thing about afrezza on the social media is I never see anyone saying they hate afrezza. The biggest issue is still the cough some get. I do see complaints from pump users about their pumps and I see many afrezza users who were pump users saying they would never go back. Clearly we are not going to agree on "how great" pumps are. So I am right except where they get the coding wrong? That mistake will be corrected pretty fast when the claim is bounced as incorrectly coded. Give Afrezza a chance, there are a few orders of magnitude more pump users than there are Afrezza users so it's unsurprising there are more pump complaints.  |
|
|
|
Post by agedhippie on Feb 19, 2023 12:37:26 GMT -5
They should highlight it on the earnings call because it's a bright spot. Is that more important than obfuscation (for competitive reasons) at this point in time? Serious question. I would expect their drug lines to broadly continue dropping; Remodulin is losing sales to the Liquidia generic sold by Sandoz, Adcira has been dropping ever since it came off patent, and Orenitram (oral treprostinil) is going nowhere. The only growth area is Tyvaso so they need to push that or risk falling back to the $100 support line. But just remember that I am famously bad at picking stocks... |
|
|
|
Post by agedhippie on Feb 19, 2023 9:29:27 GMT -5
UTHR will probably report only total revenue for Tyvaso (sum of nebulizer and DPI). I'm wondering how will we know if DPI has knocked it out of the park. I guess if total Tyvaso doubles in Q4, or something dramatic and unlikely like that, we would know. But what about 25% increase in total Tyvaso, what could we infer from that? They should highlight it on the earnings call because it's a bright spot. |
|
|
|
Post by agedhippie on Feb 18, 2023 19:05:30 GMT -5
Any bets on MNKD price action on Feb 22nd? We have to assume UTHR will present great results, and therefore the pessimistic side of me says the shorts will pour it on to keep a lid on and avoid a breakout. It will be a day to watch naked shorting. Nobody needs to naked short, there is a ton of stock available to borrow so they can simply short if they want. That said, my suspicion is that it all depends on the UTHR call. If DPI has knocked it out of the ball park I expect MNKD to rise smartly. If DPI is in line with projection then maybe a small bump as that's already priced in. |
|
|
|
Post by agedhippie on Feb 18, 2023 18:57:17 GMT -5
... In fact, it may solve one of the issues afrezza has with some people who report afrezza stops working or they need more and more. These people are building up antibodies to human insulin. Some add some subq RAA others stop using afrezza all together. For some reason the immune system is not attacking the analog. Now when it comes to convenience nothing beats afrezza. I doubt you will find many pump users who say their pumps are convenient. What's convenient having to load a new V-Go daily with insulin and then pasting it on and wearing that buldge all day and then guess how many times to push the button when eating? Lets not forget what Mike said about V-Go. When they had 80 sales reps they had an ceiling of $10M in sales. The advantage of V-Go was it was a way to get Medicare Part B to pay for the RAA. As Mike said the passage of the Inflation Reduction Act caught them by surprise and now they have $35 Part D afrezza and Mike has a board mandate to make afrezza and V-Go breakeven by year end and he already said afrezza was. I don't know why Afrezza would stop working for people, but if it was due to antibodies adding RAA wouldn't fix the problem. Sometime you just go through phases of needing more insulin for no good reason, and sometimes insulins just tail off. I have seen this happen when RAA arrived and some people had to stay on animal insulin, the same happened with Fiasp. Nobody really knows why. The new pumps are incredibly convenient, you don't have to touch them between meals, you don't have to watch you CGM to see if you need more insulin you can just let the system look after everything without bothering you. That aspect is unbeatable. I have absolutely no idea why anyone would use the V-Go - for me it's the worst of all worlds as I have something glued to me while still having to to all the work (and the work had better come in even numbers of units!) I don't think the V-Go was ever Part B since it's not DME as it's disposable. All the references I can find say V-Go is covered under Part D. I could be wrong, but I though a device had to be covered by Part B for free insulin. Has it ever been stated that insulin was free with the V-Go? |
|
|
|
Post by agedhippie on Feb 17, 2023 18:33:56 GMT -5
... IBB holds 11 million shares. Thanks for that link! So for IBB is there no stock price minimum to be included in that ETF? What about other funds, do they have minimum PPS? The ETF holding are defined by the tracker. IBB tracks the stocks in the biotech sector so they buy every stock in that sector. The number is set by the rules of the ETF,; some want equal dollar weighting, others want a percentage that reflects their value market cap in the sector. For these funds PPS is unimportant and they are purely mechanical purchasers. |
|
|
|
Post by agedhippie on Feb 17, 2023 15:41:51 GMT -5
^This is correct. There is no way that their risk officers would allow they to maintain a significant position for any length of time because; (a) this opens them up to insider trading prosecutions since as market makers they have material non-public information on the stock, (b) their regulators would stamp on their fingers in the annual audit and failing an audit is an existential risk, and (c) they are simply not staffed for it, it's not what their people are trained for (think of it like a brain surgeon doing open heart surgery - they could probably do it but it may well end badly and this is why you have specialties.) So I guess all these companies are making up stories about naked shorting?... ... You took a reply about market makers specifically and generalized it to the entire market. I did not say deliberate naked shorting didn't happen in the broader market. |
|
|
|
Post by agedhippie on Feb 17, 2023 15:37:56 GMT -5
Because it's a relatively high beta stock. Look at a plot of UTHR overlayed by MNKD and you will see they essentially track each other, but Mannkind's swings are larger (the beta.) These reports are pretty meaningless IMHO because they are simply the short position mid month and month end. They could take a huge short position on the first trading day of the month and provided closed it before the 15th it would not be reported because the reports are point in time. What you see is the short interest on those two days, not how long they have been open, sizes, who holds them, nothing useful. Thanks! Why the range of "days to cover" from 4 to 11 in the past few months if opening and closing positions can occur with frequency? I've always believed the "days to cover" was of great significance, especially if a run were to occur....which by the way, I wouldn't mind seeing next week  If I could answer that I would be a lot richer. The market is not always rational contrary to what people think (hence Keynes famous quote, “Markets can stay irrational longer than you can stay solvent.") Often all it takes is someone to build a trend and the trend followers pile in, then the trend ends and you get a reversion to mean - that happens in both directions. A run would be nice  |
|
|
|
Post by agedhippie on Feb 17, 2023 10:19:54 GMT -5
With that said, can someone either explain or even guess as to why there are over 34M shares short as of Jan 31? Because it's a relatively high beta stock. Look at a plot of UTHR overlayed by MNKD and you will see they essentially track each other, but Mannkind's swings are larger (the beta.) These reports are pretty meaningless IMHO because they are simply the short position mid month and month end. They could take a huge short position on the first trading day of the month and provided closed it before the 15th it would not be reported because the reports are point in time. What you see is the short interest on those two days, not how long they have been open, sizes, who holds them, nothing useful. |
|
|
|
Post by agedhippie on Feb 17, 2023 9:50:12 GMT -5
Since theoretical losses are unlimited with shorting, there can never be enough collateral to cover all risk. Short positions have led to the ruin of many. Market making firms would rarely allow their open holdings to get so skewed, long or short, to pose that sort of risk. Have no idea if there would be regulatory limits, but their normal mode of making money (the difference between bid and ask), or betting on volatility reversion if trading options, has much better risk-reward profile. ^This is correct. There is no way that their risk officers would allow they to maintain a significant position for any length of time because; (a) this opens them up to insider trading prosecutions since as market makers they have material non-public information on the stock, (b) their regulators would stamp on their fingers in the annual audit and failing an audit is an existential risk, and (c) they are simply not staffed for it, it's not what their people are trained for (think of it like a brain surgeon doing open heart surgery - they could probably do it but it may well end badly and yhis is why you have specialties.) |
|